Experimental Study on Mechanism of Depressurizing Dissociation of Methane Hydrate under Saturated Pore Fluid
(College of Construction Engineering, Jilin University, Changchun 130026)
Experimental Study on Mechanism of Depressurizing Dissociation of Methane Hydrate under Saturated Pore Fluid
Sun Youhong; Su Kai; Guo Wei; Li Bing; Jia Rui
(College of Construction Engineering, Jilin University, Changchun 130026)
Sediment-hosted hydrate reservoir often contains saturated pore fl uid, which changes the heat transfer and mass transfer characteristics of the hydrate reservoir. The exploitation of hydrate under saturated pore fl uid using depressurization is simulated experimentally to investigate the in fl uence of particle size of porous media, dissociation temperature, pressure drop and injected fl uid type on gas production behavior. Homogeneous methane hydrate was fi rstly formed in frozen quartz sand. With the formed hydrate sample, hydrate dissociation experiments by depressurization were conducted. The test results showed that the gas production rate of hydrate under saturated pore fl uid was substantially in fl uenced by the particle size, the pressure drop and the injected fl uid type, while it was in fl uenced little by the dissociation temperature. The hydrate dissociates faster under larger pressure drop and in the presence of smaller porous media within the experimental region. The dissociation rate increases with an increasing fl uid salinity in the initial stage, while it decreases in the later stage. The increase of gas diffusion resistance resulted from ionic hydration atmosphere in saturated chloride solution impeded the dissociation of hydrate. It can be solved by increasing the pressure drop and decreasing the fl uid salinity in the process of gas recovery from hydrate reservoir.
natural gas hydrate; porous media; saturated fl uid; depressurization; salt
1 Introduction
Natural gas hydrates are naturally occurring ice-like solids composed of water and hydrocarbon molecules, which exist in enormous quantities in the permafrost and offshore environments[1]. Because the gas hydrates contain a large amount of methane gas, hydrates in those regions have been considered to be a future energy resource with a great potential prospect[2]. Thus, the methods for developing efficient and safe exploitation of methane gas from natural gas hydrate have enormous economic and strategic significance. Four common methods of gas production from hydrate have been presented[3-4], viz.: depressurization, inhibitor injection, thermal stimulation and CO2replacement. However, each method has its own merits and demerits to some extent.
Relatively speaking, depressurization is considered to be the most economical and feasible method[5]. Yousif, et al.[6-7]measured the gas production from hydrate in Berea sandstone cores using the depressurization method. The results showed that the factors affecting depressurization included saturation, distribution uniformity and the initial permeability. Sung, et al.[8]experimentally studied the formation and dissociation of hydrate in porous media and analyzed the gas and water production dynamics during the dissociation process. Lee, et al.[9]carried out analogous experiments and found out that when the pressure drop was too large (resulted from the decrease in fi nal gas production), pores would be blocked by the regrown hydrate. Hao, et al.[10]simulated the gas production of hydrate dissociation using depressurization in an onedimensional tubular model. Under the experimental conditions, gas production rate was high, whereas the water production rate was small and relatively stable. Yang, et al.[11]conducted depressurization experiments of gas hydrate formed by ice in three-dimensional porousmedia. The dissociation occurred in the entire hydrate zone and was affected by heat and mass transfer in various stages. In these studies, the fl uid saturation was medium or low, relatively. In fact, the hydrate reservoir contains a large amount of saturated pore fl uid, especially for the case of marine hydrate reservoir[12-13]. The fl uid fi lls in the pores and porous channels of the sediment, through which the gas generated by hydrate dissociation must spread out. According to the law of diffusion, the difference in the diffusion coef fi cients between gas and liquid can be up to four orders of magnitude. Furthermore, the heat transfer of the gas hydrate reservoir is also different due to the relatively large difference in thermal conductivity between them. In this work, a formation method for gas hydrate was proposed to obtain an ideal hydratesediment sample. Then, the dissociation experiments via depressurization under saturated pore fl uid had been conducted with the formed hydrate-sediment sample. These results are expected to provide some theoretical guidance for the exploitation of natural gas hydrate under saturated pore fl uid.
2 Experimental
2.1 Experimental apparatus and materials
A schematic of the experimental apparatus is shown in Figure 1, which is divided into four parts, namely: the reaction system, the injection system, the gas flow detection system and the data collection system. The primary component of the reaction system is the hydrate formation/dissociation reactor (with a volume of 254.5 cm3), which can withstand a pressure of higher than 20 MPa. The reactor is enclosed in a water-bath that can be maintained at a constant temperature with a precision of 0.1 ℃. A resistance thermometer (Pt100) and a pressure transducer (PTX1517) inserted into the reactor are used to measure the temperature and pressure of the system. The accuracy of the resistance thermometer and pressure transducer is ±0.2 ℃ and ±0.02 MPa, respectively. The main part of the injection system is a hand pump, the maximum working pressure of which can be up to 30 MPa. The gas flow detection system includes a back-pressure regulator, a filter and a gas flowmeter. The pressure and temperature data are collected by the corresponding transducers in real time over the whole experimental period, and the fl ow rate data are calculated arti fi cially.
The methane with a purity of 99.99% used in the experiments was supplied by the Changchun Beifang Gas Industry Corporation. Sodium chloride had a purity of higher than 99.5%. Distilled water was used to prepare all solutions. The quartz sand with a purity of 99.0% was cleaned and dried before each experiment.
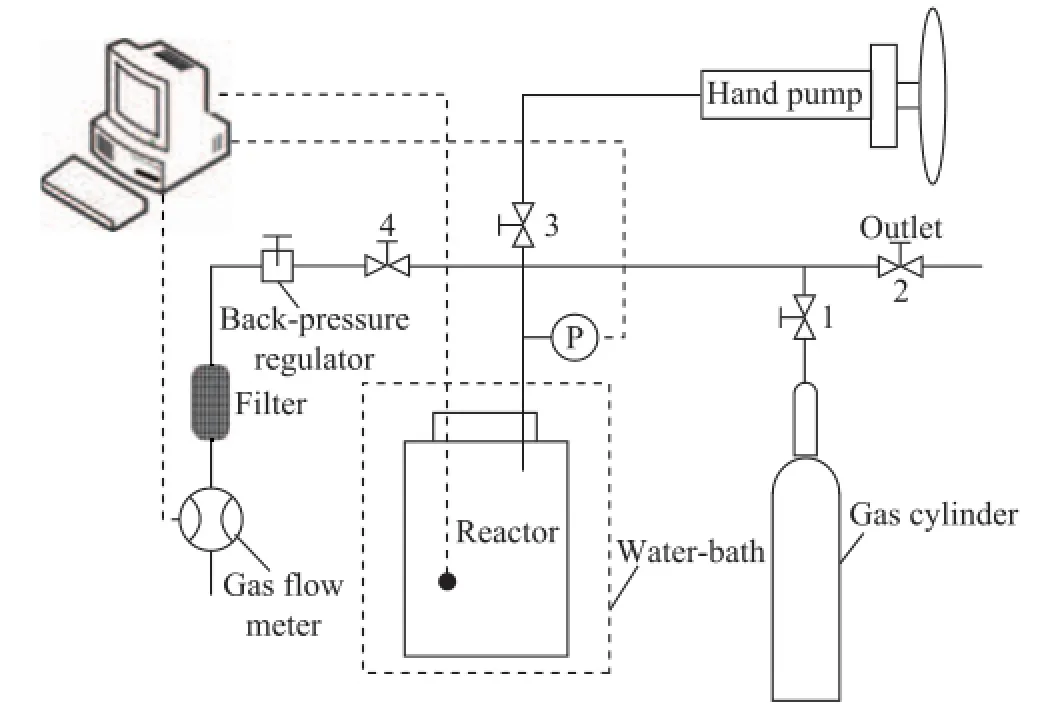
Figure 1 Schematic diagram of the experimental apparatus
2.2 Experimental
2.2.1 Ideal hydrate formation
To investigate the distribution of dissociation rate over the whole sediment hydrate sample, as well as its evolution with elapsed time when the sample was surrounded with fluid under reservoir pressure conditions, the original hydrate was demanded to distribute in the sediment homogeneously. Before the experiment, the reactor was washed with distilled water and dried. After that, 15 g of distilled water and 155 g of quartz sand were both weighed precisely and frozen. Then, the distilled water was mixed with the frozen sand immediately and adequately. The reactor was arranged in a waterbath, the temperature of which was set to -6 ℃ to make the distilled water freeze onto and enwrap the surface of the quartz sand. After the system temperature became stable, methane gas was injected into the reactor at 6 MPa, and the water-bath temperature was gradually raised to 0.5 ℃to promote the hydrate formation. When the temperature and pressure changes were negligible, it is assumed that the hydrate formation was completed.
2.2.2 Hydrate dissociation via depressurization
The hydrate dissociation test started after the representative sediment hydrate sample was formed. At first, the water bath was preheated to the experimental temperature and the back-pressure regulator was adjusted to the experimental pressure. Valve 2 was opened adequately, and the system pressure declined gradually until it reached a surplus pressure of 0.2 MPa above the equilibrium pressure of methane hydrate at the corresponding temperature. Subsequently, a wet towel precooled to the experimental temperature was wrapped around the hand pump. A certain amount of distilled water or fluid which was frozen in advance to avoid hydrate dissociation was injected into the reactor rapidly by opening the valve 3. After that, valve 3 was closed and valve 4 was opened quickly to make the system depressurized to the set pressure. Then, the gas flow detector was connected to the system. When the flow rate was less than 10 mL/min, valve 4 was closed and the gas production was considered to be fi nished. During the whole experimental period, the values of temperature, pressure, and the gas fl ow rate were all recorded by the computer.
3 Results and Discussion
3.1 Formation of the sediment hydrate samples
Because the experiments were focused on the dynamics of the dissociation of the hydrate samples and the uniformity of which had a substantial influence on the test results[7], the original hydrate was demanded to distribute in sediment homogeneously. However, it has been demonstrated that water might migrate in sediment due to capillary effect[14-15]and the hydrate could mainly form in the vessel wall and the upper section of the sediment as shown in Figure 2(a). Contrarily, Figure 2(b) shows the hydrate sample formed from frozen quartz sand according to the method described in section 2.2.1. The sand bed with hydrate was found to be consolidated, and no discernible wall-climbing phenomena could be found. To confirm the existence of hydrate inside the consolidated sediment, a certain amount of water at room temperature was poured into the reactor. The phenomenon is shown in Figure 2(c). We can see that the hydrate sample dissociated drastically and produced a large number of gas bubbles, indicating that the hydrate existed homogeneously in the sand sediment. In addition, to investigate the conditions of hydrate dissociation under saturated pore fl uid, the hydrate sample formed in each experiment was demanded to be analogical. Table 1 shows the parameters of hydrate sample and experimental conditions for hydrate dissociation.
3.2 Law of temperature and pressure changes
Figure 3 indicates the evolution of temperature and pressure stimulated by hydrate dissociation, where the temperature and pressure drop are 3 ℃ and 1 MPa, respectively, with the saturated pore fluid made of distilled water. The temperature and pressure changes over time can be divided into four stages, viz.:t1(0—5.6 min),t2(5.6—6.1 min),t3(6.1—24 min) andt4(24—54 min).
t1is the low-temperature fluid injection stage. After the hydrate formation was completed, the pressure was reduced to 0.2 MPa above the equilibrium pressure. After that, the distilled water frozen in advance was rapidly injected into the reactor by the hand pump. Owing to the Joule-Thomson effect, the temperature increased rapidly from 3 ℃ to 6.7 ℃, and the pressure increased from 4.1 MPato 5.2 MPa, synchronously. In addition, there was a delay in the response of the temperature compared with that of the pressure. The pressure decreased by 0.05 MPa as the temperature increased by 0.15 ℃ because of heat transfer.

Figure 2 The morphology of gas hydrate formation
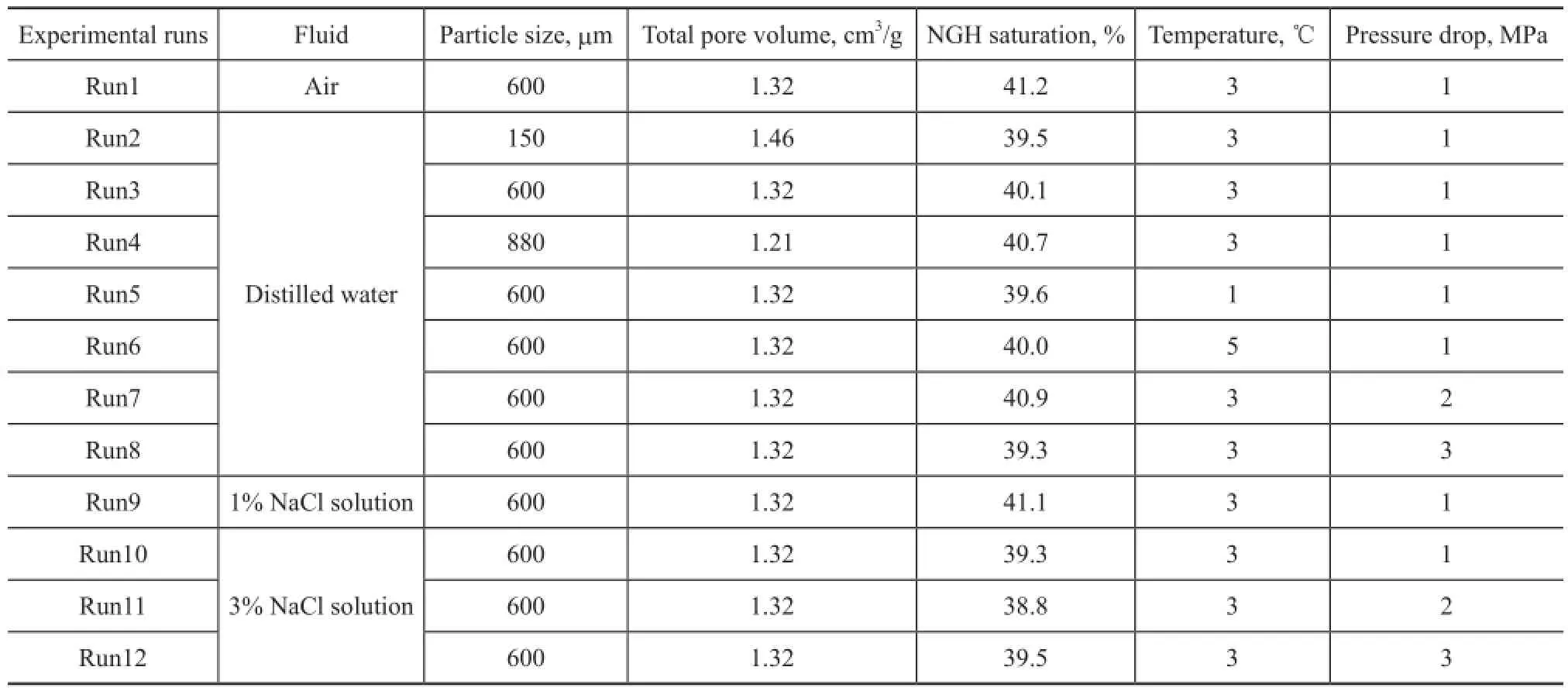
Table 1 Parameters of hydrate sample and experimental conditions for hydrate dissociation
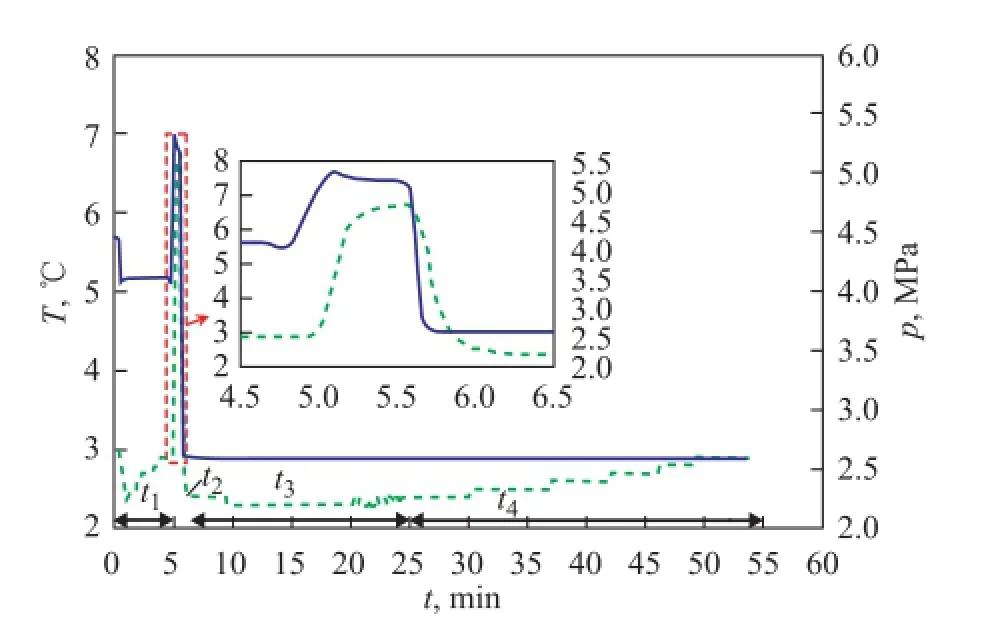
Figure 3 Temperature and pressure changes stimulated by the hydrate dissociation
t2is the pressure venting stage. After opening the vent valve, the pressure decreased quickly to 2.60 MPa, as set by the back-pressure regulator. The temperature dropped rapidly to the experimental temperature because of the endothermic reaction of hydrate dissociation process and gas throttling effect. And the latter one was the main reason. After the pressure dropped to the experimental pressure value, the temperature, which was approximately 4 ℃, continued to decline rapidly. The reason is described above.
t3is the fast dissociation stage. At this stage, a massive amount of hydrate decomposed rapidly, and the temperature continued to decline to 2.5 ℃. Then, the hydrate reached a stable dissociation stage, where a dynamic balance between the heat absorption of hydrate dissociation and heat transfer from the water-bath took place because the saturated fluid strengthened the heat transfer process greatly. Finally, the temperature fl uctuated at 2.3℃, whereas the pressure was stable in this period.
t4is the final dissociation stage. The dissociation rate reduced, and the heat absorbed in hydrate dissociation was less than the heat transferred from the water-bath. Owing to this, the temperature rose faster over time. Finally, the temperature was equal to the experimental temperature. This stage was the longest one.
3.3 Law of gas production
3.3.1 Effect of particle size of porous media on gas production
In order to clarify the in fl uence of porous medium on the gas production under saturated fl uid, the experiments of methane hydrate dissociation were conducted in a porous medium made of quartz sand with different particle size, such as 150 μm, 600 μm, and 880 μm. The results are shown in Figure 4. In this study, we found out that the particle size of quartz sand used as the porous medium had an insignificant influence on gas production rate. According to the experimental results, the largest gas production rate of methane hydrate was 1 536 mL/min in the porous medium with a particle size of 150 μm, while it was 1 130 mL/min and 1 023 mL/min in the porousmedium with a particle size of 600 μm and 880 μm, respectively. The results showed that the smaller the particle size was, the faster the methane hydrate could dissociate in the initial stage. In Figure 4, the whole time of gas production of methane hydrate in the porous medium with a particle size of 150 μm was less than that in the porous media with a particle size of 600 μm and 880 μm, respectively. Since the porous medium with different particle size had different speci fi c surface area, so it would result in different dissociation rate of methane hydrate. In addition, the smaller the particle size of porous medium was, the bigger the speci fi c surface area would be, which was bene fi cial to the process of gas production in the system of hydrate dissociation. And less time was taken to fi nish the dissociation process.
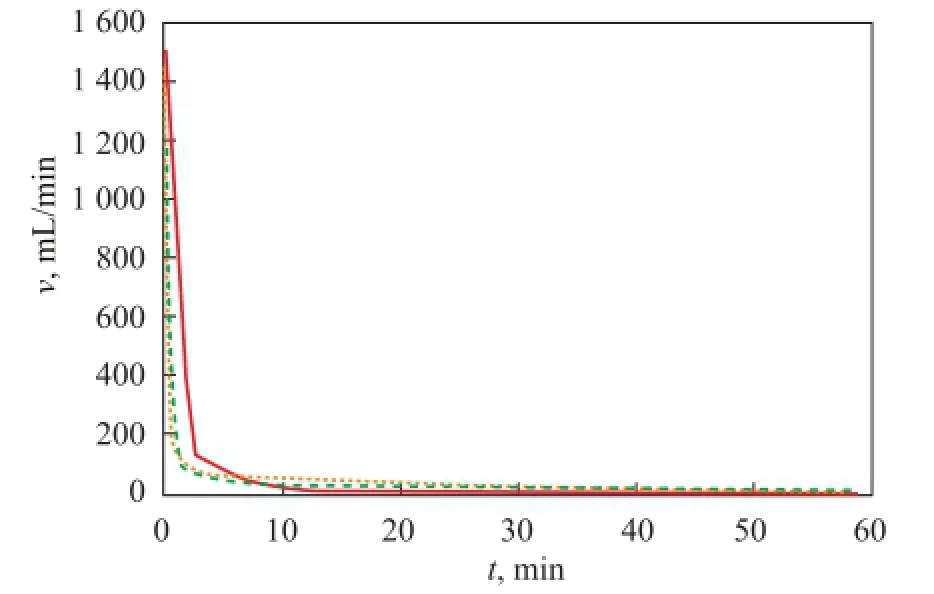
Figure 4 Gas production rate changes at particle size of porous media
3.3.2 Effect of temperature on gas production
The evolution of the gas production rate for runs 3, 5 and 6 at different temperatures is shown in Figure 5, in which the pressure drop was 1 MPa with the saturated pore fl uid made of distilled water. The gas production rate curves of the hydrate at the three temperatures was basically identical, and the initial gas production rate, which was much higher than that in the late stage, was the highest. By taking the run 3 as an example, the gas production rate was up to 1 130 mL/min in the initial stage of dissociation, whereas it declined quickly to 200 mL/min. Afterwards, it decreased slowly to a minimum of 10 mL/min at 48 min. When the gas fl ow rate was lower than 10 mL/min, the gas could not be measured due to the low precision of the gas fl owmeter. Kim, et al.[16]believed that the dissociation rate of hydrate is controlled by the first-order kinetics and it is proportional to the surface area and to the driving force, which can be expressed by the following formula.
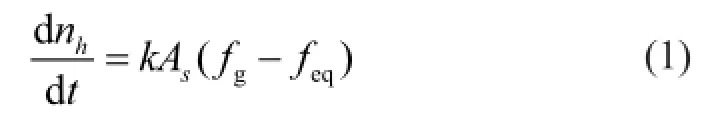
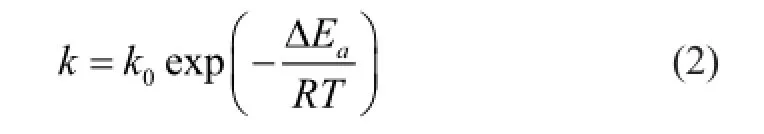
wherek0is the basic reaction kinetics constant, ΔEais the activation energy,Ris the gas constant (8.314 J/mol·K), andTis the temperature (K).
According to Eq. (1),Asandfg-feqcan be considered to be unchanged in the experiments. The dissociation rate is only in fl uenced byk, which also can be considered to be unchanged, relatively, because its unit is expressed in degrees Kelvin and the largest gap in temperature in each group is only 1.46%. Therefore, it is concluded that, within a certain experimental temperature range, the temperature has little effect on the gas production rate of the hydrate surrounded with saturated pore fl uid.
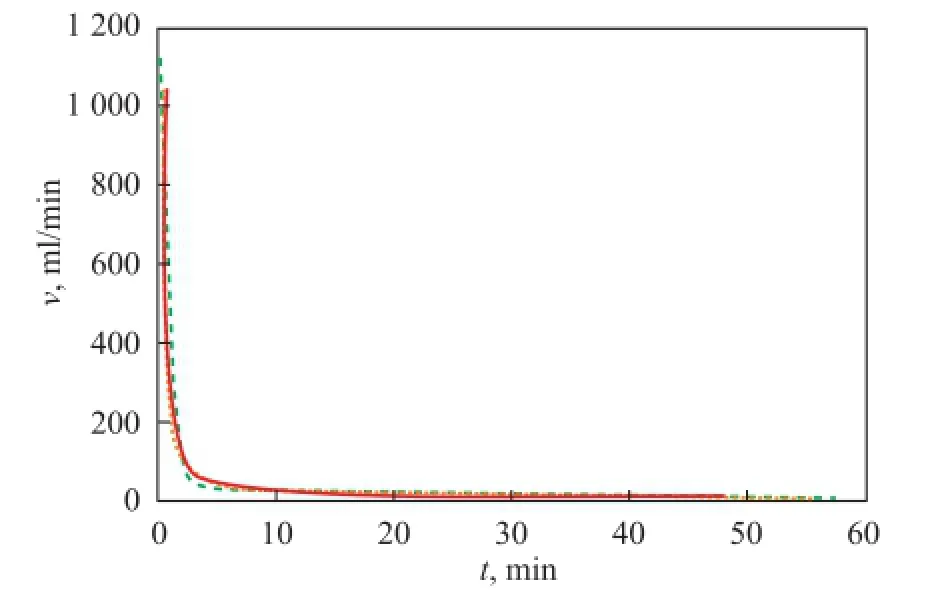
Figure 5 Gas production rate changes at different temperatures
3.3.3 Effect of pressure drop on gas production
The pressure drop is usually regarded as the main driving force during the depressurization process. To evaluate the effect of pressure drop on the real gas production, a series of experiments on pressure drop have beenconducted. Figure 6 shows the variation of gas production rate with time from hydrate dissociation for runs 3, 7, and 8, where the temperature was 3℃ and the saturated pore fluid was made of distilled water. It can be seen that the gas production rate increased with the increase of pressure drop. In the initial stage, the gas production rate could be maintained momently at a valuev0, andv0increased with an increasing pressure drop. After that, the gas production rate declined dramatically. When the pressure drop reached 1 MPa, the initial gas production rate was 1 300 mL/min, and thev0was 380 mL/min. Furthermore, when the pressure drop reached 2 MPa and 3 MPa, respectively, the initial gas production rate was 1 580 mL/min and 1 870 mL/min, respectively, while the value ofv0was 770 mL/min and 984 mL/min, respectively. This occurred because a part of the hydrate had been dissociated in the pressure venting period. After the flowmeter was connected to the system, the partial free gas was not evacuated, and the previously dissociated gas was measured along with the gas produced from depressurization together. In addition, with the increase of pressure drop, the total time for hydrate dissociation decreased and the whole dissociation rate increased. In particular, when the pressure drop was 1 MPa, the total dissociation time was 49 min. However, when the pressure drop was 2 MPa and 3 MPa, respectively, the total dissociation time was equal to only 15.4 min and 9.3 min, respectively. It is evident that the time used for exploiting the hydrate reservoir under saturated pore fl uid using the depressurization method decreased as compared with the normal hydrate reservoir.
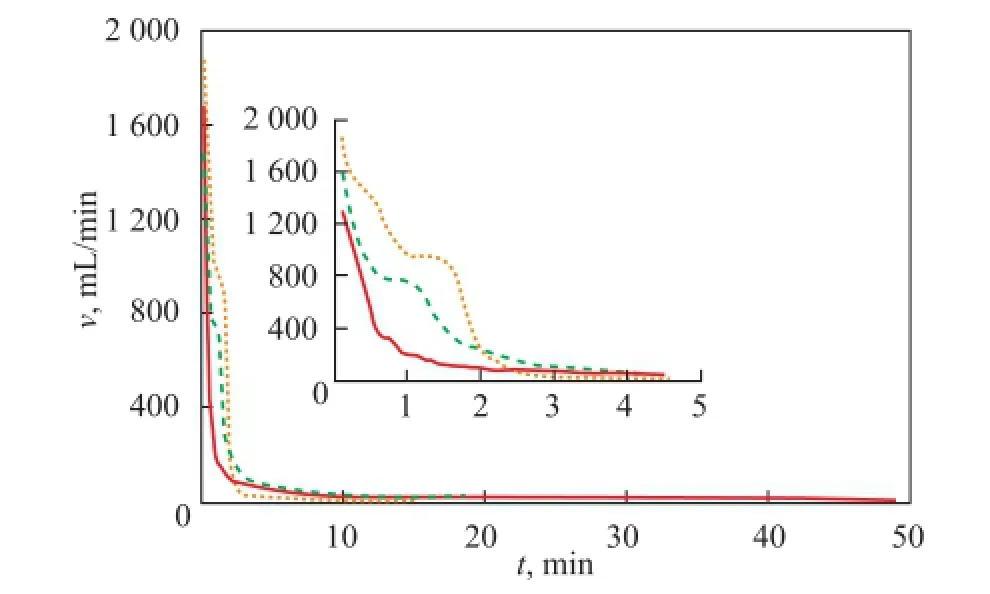
Figure 6 Gas production rate changes at different pressure drop values
In order to simulate the marine salt environment, the gas production experiments for runs 10, 11 and 12 in the presence of 3% NaCl solution were conducted. Figure 7 depicts the variation of gas production rate at different pressure drop values. It implies that the gas production rate increased with an increasing pressure drop, which was similar to the above-mentioned case. However, the gap between the initial gas production rate and the stable gas production rate was smaller. The reason was that the hydrate dissociated faster in the presence of NaCl solution, which weakened the effect of pressure drop. Furthermore,v0fluctuated, which was different to that for the case with distilled water. The dissociation frontier continued to extend and infiltrate during the whole dissociation process. The salt interacted with the hydrate at random with the gas produced unstably. However, the hydrate dissociated rapidly just for 2.6 min and stopped, which went contrary to the law. When the gas fl ow was under 10 mL/min, the reactor was opened and a block of hydrate was found as shown in Figure 8. The experiments were conducted repeatedly, and similar results were obtained.
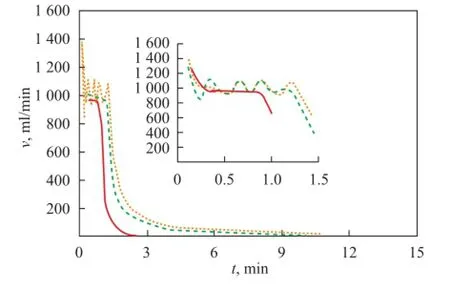
Figure 7 Gas production rate of the hydrate surrounded with 3% NaCl solution at different pressure drop values

Figure 8 Hydrate does not dissociate in 3% NaCl solution
3.3.4 Effect of fl uid type on gas production
Figure 9 shows the cumulative gas productions of runs 1, 3, 9 and 10 with different saturated pore fl uids, while the temperature and pressure drop were 3℃ and 1 MPa, respectively. The period from the beginning of gas production to a gas production rate of 200 mL/min is namedt0. The gas accumulated in this period is denoted asV0, and the ratio betweenV0and the total amount of produced gas is calledη. As shown in Figure 9,t0,V0andηall increased successively in an increasing order of the air, the distilled water, the 1% NaCl solution and the 3% NaCl solution, respectively, in the initial stage. The law can also be expressed as follows: the dissociation rate increased with an increasing salinity of the fluid. The speci fi c data are shown in Table 2. However, in the later stage, the law of gas production in different pore fl uids is contrary to that applicable to the initial stage.
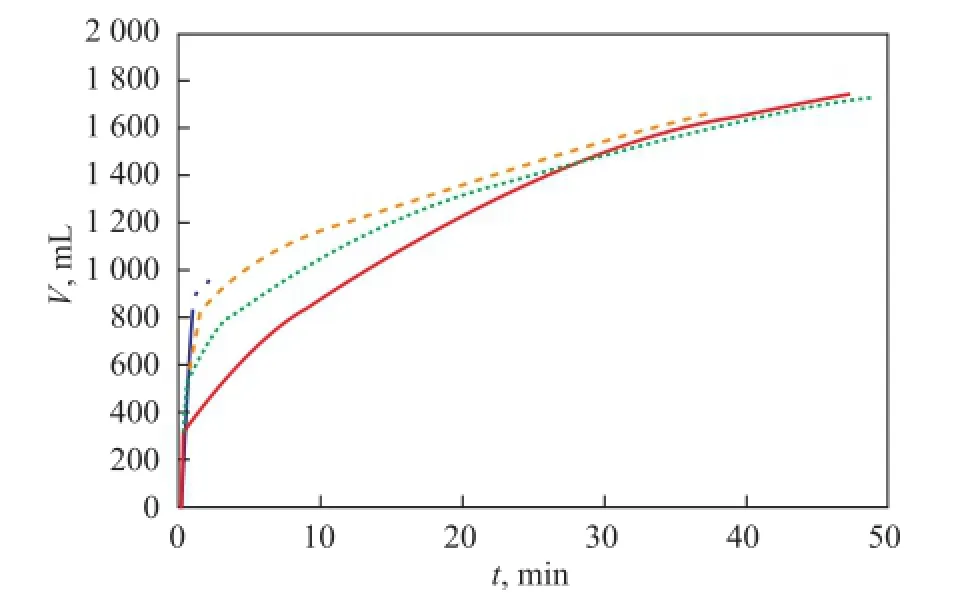
Figure 9 The amount of gas accumulated in different pore fl uids

Table 2 Hydrate dissociates under different saturated pore fluids in the initial stage
Methane hydrate is an inclusion compound where hydrogen-bonded water molecules encage appropriate guest molecules under high pressure and low temperature conditions, the dissociation of which was performed during the destruction of the hydrate lattice and the release of gas on the surface of the hydrate. Naslund, et al.[17]used X-ray absorption spectroscopy (XAS) and X-ray Raman scattering (XRS) technique to measure the O edge XAS and XRS spectra in the presence of pure water and 1 mol/L of NaCl solution. The results showed that Na+ions could break or weaken the hydrogen bond network of water molecules. Soper, et al.[18]studied the in fl uence of ions on water structure in a salt solution using the isotope substitution neutron diffraction technique. They found out that negative and positive ions appreciably damaged the water structure. Consequently, upon injecting the salt solution, salt ions which were in contact with the hydrate cages could weaken the hydrogen bonds between water molecules, resulting in the destruction of hydrate cages and release of methane gas. The dissociation of the hydrate was accelerated with the increase of salinity. In addition, the heat transfer of the system differed in different fl uids based on the Fourier law.

In the later stage, according to the Fick’s law of diffusion, the following equation is obtained:

whereis the density of molar flow,Cis the total molar concentration,DABis the diffusion coefficient, andis the concentration gradient of component A in theXdirection. The difference between the diffusion coef fi cients of gas in the gas and liquid media can reach four orders of magnitude. The reason is that, compared with the molecular diameter of gas, the distance between gas molecules is very large, as evidenced by the small molecular binding force and diffusion resistance. However, the molecular distance of the liquid is small relative to its diameter. Liquid molecules can collide with the neighboring ones frequently in diffusion,so that the diffusion resistance is large. Hence, the diffusion resistance of hydrate under saturated pore water is much larger than that in gas during dissociation. Furthermore, based on the liquid solution theory, NaCl, as a strong electrolyte, dissolves easily in water and produces a strong electrostatic fi eld, which causes water molecules with strong polarity and high permittivity to gather with Na+ions to form associated molecules. In contrast, methane molecules with weak polarity and low permittivity are displaced from Na+ions[19-20]. Gas molecules are clustered in the farther zone due to solvation effects, and the water molecules aggregate to form an ionic hydration atmosphere around the ions, which can form a methane protective tissue around the hydrate particles, as shown in Figure 10. The tissue has a self-protective effect[21], which would increase the diffusion resistance and reduce the hydrate dissociation rate. Accordingly, when the fluid and pressure drop are 3% NaCl solution and 1 MPa, respectively, the driving force is less than the mass transfer resistance, so that the hydrate dissociates much more slowly and there is an amount of un-dissociated hydrate remaining thereby. Contrarily, when the pressure drop is 2 MPa or 3 MPa, the driving force of dissociation is higher than the mass transfer resistance, and the dissociation would continue. In the later stage, the in fl uence of mass transfer is greater than that of salinity and heat transfer. The law of hydrate dissociation in different saturated pore fl uids is contrary to that governing the initial stage. In the process of actual exploitation of the hydrate reservoir, increasing the pressure drop and reducing the salinity of fluid can be used to solve this problem.
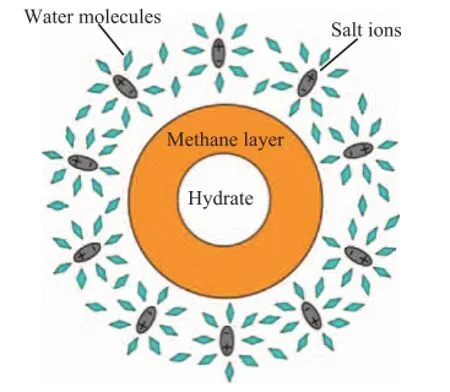
Figure 10 Hydration ion atmosphere of the salt solution
4 Conclusions
In this work, we firstly formed homogeneous methane hydrate in quartz sand based on a home-made experimental apparatus. Next, the hydrate dissociation experiments by depressurization were performed with the formed methane hydrate sample. The major fi ndings could be summarized as follows:
(1) The hydrate, which was homogeneously distributed in quartz sand, was formed successfully according to the method reported in this work. Based on this approach, the effect of the distribution of hydrate on the dissociation rate was avoided.
(2) The gas production of hydrate under the saturated pore fluid was substantially influenced by the porous media, the pressure drop and the salinity, while it was in fl uenced little by temperature. The hydrate could dissociate faster under larger pressure drop and in smaller porous media within the experimental region. The effect of fl uid salinity on gas production was contrary among the results obtained in the initial stage and in the later stage, relatively.
(3) For the fl uid type investigated, it was found out that the dissociation rate increased with an increasing fluid salinity in the initial stage, which was mainly controlled by the salinity and the heat transfer process. Yet, the law of gas production in the later stage that was controlled by mass transfer was contrary to that applicable to the initial stage.
(4) The existence of gas diffusion resistance resulted from ionic hydration atmosphere in the saturated chloride solution slowed the dissociation of hydrate. When the temperature, pressure drop and the pore fl uid were 3 ℃, 1 MPa and 3% NaCl solution, respectively, the hydrate stopped dissociating soon and there was a blockage of undissociated hydrate in the reactor. This problem could be solved by increasing the pressure drop or decreasing the fl uid salinity in the actual exploration environment.
Acknowledgements: This study was supported by the National Natural Science Foundation of China (Grant No.51304079, Grant No.51474112, Grant No.41502343 and Grant No.51506073).
[1] Sloan E D. Fundamental principles and applications of natural gas hydrates[J]. Nature, 2003, 426(6964): 353-359
[2] Zhou X T, Fan S S, Liang D Q, et al. Replacement of methane from quartz sand-bearing hydrate with carbon dioxide-in-water emulsion[J]. Energy & Fuels, 2008, 22(3): 1759-1764
[3] Zhang X M, Li J P, Wu Q B, et al. Experimental study on the characteristics of CO2hydrate formation in porous media below freezing point[J]. China Petroleum Processing and Petrochemical Technology, 2015, 17(3): 32-38
[4] Sun C Y, Chen G J. Methane hydrate dissociation above 0 ℃ and below 0 ℃[J]. Fluid Phase Equilibria, 2006, 242(2): 123-128
[5] Collett T S, Kuuskraa V A. Hydrates contain vast store of world gas resources[J]. Oil & Gas Journal, 1998, 96(19): 90-95
[6] Youslf M H, Li P M, Selim M S, et al. Depressurization of natural gas hydrate in Berea sandstone cores[J]. Journal of Inclusion Phenomena and Molecular Recognition in Chemistry 1990, 8(1): 71-88
[7] Youslf M H, Sloan E D. Experimental investigation of hydrate formation and dissociation in consolidated porous media[J]. SPE Reservoir Engineering, 1991, 6(4): 452-458
[8] Sung W M, Kang H Y. Experimental investigation of production behaviors of methane hydrate saturated in porous rock[J]. Energy Sources, 2003, 25(8): 845-856
[9] Lee J, Park S, Sung W. An experimental study on the productivity of dissociated gas from gas hydrate by depressurization scheme[J]. Energy Conversion and Management, 2010, 51(12): 2510-2515
[10] Hao Y M, Bao Q W, Chen Y M. Laboratory investigation of pressure development of natural gas hydrates[J]. Petroleum Exploration and Development, 2006, 33(2): 217-220 (in Chinese)
[11] Yang X, Sun C Y, Su K H, et al. A three-dimensional study on the formation and dissociation of methane hydrate in porous sediment by depressurization[J]. Energy Conversion and Management, 2012, 56: 1-7
[12] Hu G W, Ye Y G, Zhang J, et al. Study on gas hydrate formation-dissociation and its acoustic responses in unconsolidated sands[J]. Geoscience, 2008, 22(3): 465-474 (in Chinese)
[13] Ecker C. Seismic Characterization of Methane Hydrates Structures[D]. San Francisco: Stanford University, 2001: 79
[14] Hovland M, Gallagher J W, Clennell M B, et al. Gas hydrate and free gas volumes in marine sediments: example from the Niger Delta front[J]. Marine and Petroleum Geology, 1997, 14(3): 245-255
[15] Yang B, Li M D, Du N S, et al. Study on temperature variation of natural gas hydrate formation in gas tank[J]. Natural Gas and Oil, 2014, 32(5): 1-4 (in Chinese)
[16] Kim H C, Bishnoi P R, Heidemann R A, et al. Kinetics of methane hydrate decomposition[J]. Chemical Engineering Science, 1987, 42(7):1645-1653
[17] Naslund L A, Edwards D C, Wernet P, et al. X-ray absorption spectroscopy study of the hydrogen bond network in the bulk water of aqueous solutions[J]. The Journal of Physical Chemistry A, 2005, 109(207): 5995-6002
[18] Soper A K, Weckstr?m K. Ion solvation and water structure in potassium halide aqueous solutions[J]. Biophysical Chemistry, 2006, 124(3): 180-191
[19] Sun S C, Liu C L, Ye Y G, et al. Dissociation conditions and in fl uencing factors of methane hydrate in chloride salt solution under high pressure[J]. Acta Physico-Chimica Sinica, 2011, 27(12): 2773-2778 (in Chinese)
[20] Huang Z Q. Introduction to the Electrolyte Solution Theory[M]. Revised edition. Beijing: Science Press, 1983 (in Chinese)
[21] Li S X, Xu X H, Zheng R Y, et al. Experimental investigation on dissociation driving force of methane hydrate in porous media[J]. Fuel, 2015, 160: 117-122
Received date: 2015-12-23; Accepted date: 2016-03-18.
Professor Guo Wei, E-mail: guowei6981@126.com.
- 中國(guó)煉油與石油化工的其它文章
- Study on Preparation and Properties of Grease Based on Ultra fi ne Bentonite Powder
- Preparation and Tribological Properties of Lanthanumdoped Muscovite Composite Particles as Lubricant Additives in Lithium Grease
- Preparation and Tribological Behavior of Hydrophobic Lanthanum Borate Nanosheets in Rapeseed Oil
- Synthesis and Evaluation of Environmentally Friendly Calcium Isostearate Detergent with Excellent Oil Solubility
- Experimental and Molecular Simulations for Evaluating the Effect of Lubricity Improvers on the Property of Jet Fuel
- Analysis and Modeling of Wangqing Oil Shale Drying Characteristics in a Novel Fluidized Bed Dryer with Asynchronous Rotating Air Distributor

