Comparison of Lubricities of Two Novel Benzotriazole Derivatives Used as Additives in Water-Glycol Hydraulic Fluid
Zheng Zhe; Wang Jianhua; Fang Jianhua; Jiang Zeqi; Gu Kecheng
(1. Department of Oil, Army Logistics University of PLA, Chongqing 401311;2. No. 92228 Unit of PLA, Beijing 100072; 3. College of Pharmacy, Army Medical University,Chongqing, 400038)
Abstract: Two kinds of benzotriazole derivatives with and without sulfur (abbreviated as BSC and BC, respectively,) were synthesized and their lubricating, anticorrosion and antirust properties were investigated, when they were used as additives in the water-glycol fluid. The morphology and chemical species of typical elements on the worn surface were examined by the scanning electron microscopy (SEM), the energy dispersive spectrometry (EDS), and the X-ray photoelectron spectroscopy (XPS) in order to reveal the tribological mechanisms. The results indicated that two synthesized additives could effectively improve the anti-wear, friction-reducing, load-carrying, anticorrosion, and antirust properties of the base fluid. The surface analysis illustrated that stable nitrogen-containing absorption films generated by BC should be responsible for its facilitated anti-wear and friction-reducing performance, and the excellent lubricities of BSC should be attributed to the combined action of adsorption film and tribo-chemical film which were composed of iron oxides, iron sulfides and iron sulfates.The superior lubricating properties of BSC as compared to that of BC demonstrated the effectiveness of elemental sulfur in reducing friction and wear, especially under high loads. But corrosion wear is more obvious at a relative high concentration of elemental sulfur.
Key words: hydraulic fluid; water-soluble additives; benzotriazole; lubricating properties
1 Introduction
In hydraulic systems, fire safety is an important factor which should be considered because of the high ambient temperature[1]. It has been known that the waterglycol fluid, which possesses several advantages, such as fire resistance, low cost, excellent stability, and environmental-friendly property, is one of the most common alternatives to mineral oil when it is employed in such systems[2-3]. In general, about 40% by volume of water are added into the water-glycol fluids to achieve good fire-resistant property and cooling ability[4-5].However, the lubricating properties of water-glycol hydraulic fluids, which determine the reliability and availability of the hydraulic system, are not usually satisfactory, mainly due to the inability of water to form an effective film to reduce friction and wear in the elastohydrodynamic lubrication and boundary lubrication environments[6-8]. This disadvantage greatly limits the use of water-glycol hydraulic fluids in severe industrial applications[9-10].
In the previous studies, various water-soluble lubricant additives which could form tribological films through physical or chemical adsorption, deposition, and tribochemical reaction, were added into the water-based fluids to improve their lubricity. Among these additives, the long-chain carboxylic acid has been demonstrated to be a most useful ingredient thanks to their moderate prices and easy disposal[11-12]. Nevertheless, it also has been proved that the adsorption films formed by the carboxylic acids would be destroyed under some severe conditions because there are no “active elements” in carboxylic acids to generate effective tribo-chemical films at high pressure contacts[13-14]. Nowadays, many researches have reported that the nitrogen-containing heterocyclic compounds,such as triazine, benzothiazole and thiadiazole derivatives,have excellent extreme-pressure property when they are employed in the oil-based lubricants mainly because of their “active elements” and compact structures[15-18].Unfortunately, their solubility and the ability to generate competitive adsorption film on metal surface in water could not meet expectation. Thus, there is an urgent need for further design of these compounds to improve their performance in the water-glycol hydraulic fluids.
Benzotriazole (abbreviated as BTA), a kind of nitrogencontaining heterocyclic compound, is usually used as antirust agent and metal passivator in lubricating oils[19].On the other hand, its derivatives with elemental sulfur or phosphorus can exhibit satisfactory anti-wear and friction-reducing properties in severe conditions[20-21].However, most of the current researches only focus on their corrosion inhibiting performance but neglect their lubricating properties when they are employed as water-soluble additives. In this paper, two novel BTA derivatives (abbreviated as BC and BSC) combining alkyl carboxyl group with BTA molecule, were synthesized based on the concept of molecular design. Particularly,elemental sulfur was added in BSC to investigate its effect on the performance of water-glycol hydraulic fluid.Their friction and wear characteristics functioning as additives in water-glycol fluids were evaluated and the possible tribological mechanism was also analyzed. In addition, their copper corrosion-inhibiting property and antirust property were investigated for the potential use in practical application.
2 Experimental
2.1 Synthesis and structural analysis of BTA derivatives
The analytical reagent grade chemicals (benzotriazole,carbon disulfide, sodium hydroxide, 11-bromoundecanic acid), and deionized water were used to synthesize 1H-benzotriazole-1-undecylic acid (abbreviated as BC), and S-(11-carboxylundecyl)-1-(1H- benthiazolyl)dithioformiate (abbreviated as BSC). The routes for synthesis of the additives are presented in Figure 1.
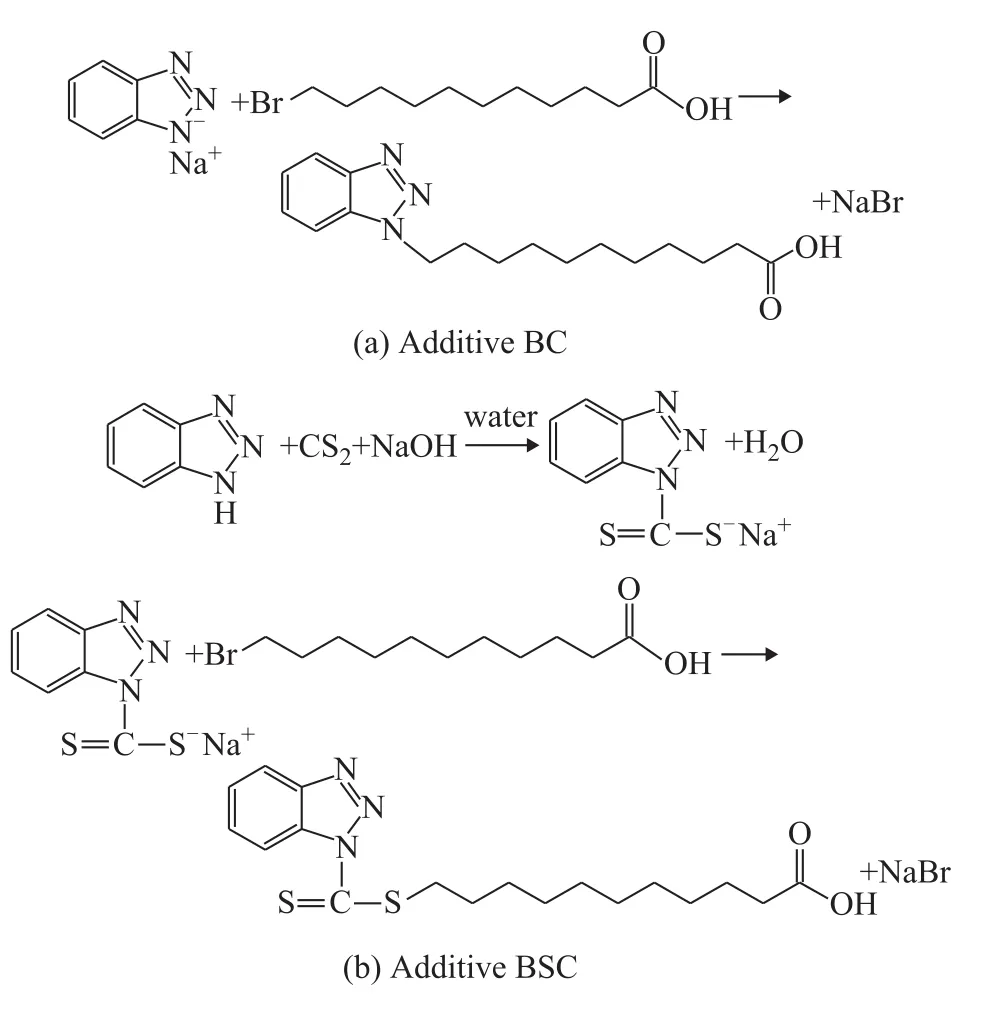
Figure 1 Routes for synthesis of benzotriazole derivatives
The detailed specific process is as follows: 0.1 mol of benzotriazole, 60 mL of deinoized water, and 0.1 mol of NaOH were added into a 500-mL three-necked flask under stirring at room temperature for 1 hour to obtain the sodium benzotriazole solution. Next, 0.1 mol of 11-bromoundecanic acid was added into a 250-mL beaker, and then a 0.1 mol/L sodium carbonate solution was slowly added into the beaker under vigorous stirring until the solution turned transparent to yield the sodium 11-bromoundecanic acid solution. Afterwards, the sodium 11-bromoundecanic acid solution was slowly added into the sodium benzotriazole solution under stirring at room temperature, then the mixture was heated up to 90 °C and refluxed for 6 hours. After being cooled down to room temperature, a large amount of white precipitate appeared by acidizing the solution with concentrated hydrochloric acid until the pH value was equal to 1.0,and the precipitate was obtained by suction filtration. The precipitate was rinsed with deinoized water repeatedly until no chloridion was detected. Finally, white solid BC was obtained after removing the water through vacuum drying.
The synthesis of BSC containing sulfur was similar to that of BC, albeit via more complicated procedures. At 15 °C, 0.11 mol of carbon disulfide was slowly dropwise added in the sodium benzotriazole solution, which was prepared in the same way with the same quantity of reagents mentioned above, followed by stirring at room temperature for 8 hours to obtain the yellow sodium 1-(1H-benthiazolyl) dithioformiate solution. Next, the sodium 11-bromoundecanic acid solution was slowly added into the sodium 1-(1H-benthiazolyl) dithioformiate solution, and then the mixture was heated up to 90 °C and refluxed for 6 hours. After acidizing the mixture with concentrated hydrochloric acid, a large amount of yellow precipitate appeared. The precipitate was filtered and rinsed with deinoized water repeatedly until no chloridion was detected. At last, the yellow solid BSC was obtained by removing the water through vacuum drying.
The chemical structure of these benzotriazole derivatives was characterized by the Fourier transform infrared spectroscopy (FT-IR) and elemental analysis. The FT-IR spectra of BC and BSC are shown in Figure 2.
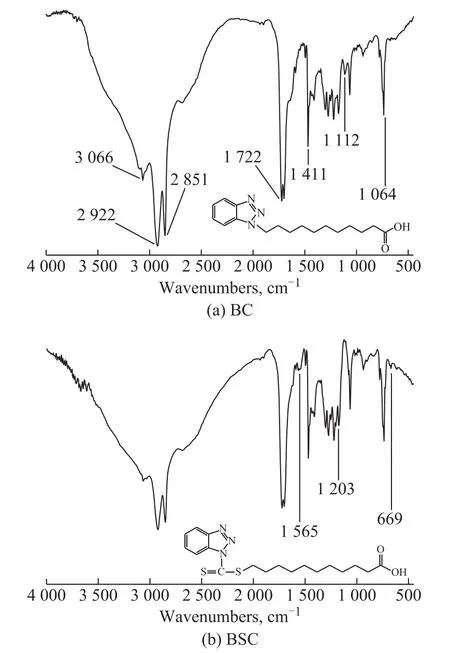
Figure 2 FT-IR spectra of the synthesized benzotriazole derivatives
The FT-IR spectra of BC (KBr) are identified at:3 066.20 cm-1(O-H stretch vibration); 2 922.64 cm-1and 2 851.43 cm-1(C-H stretch vibration); 1 722.62 cm-1(C=O stretch vibration); 1 589.55 cm-1and 1 495.06 cm-1(benzene ring stretch vibration); 1 411.09 cm-1(N=N stretch vibration); 1 301.98 cm-1(arylamine C-N stretch vibration); 1 112.75 cm-1(C-N stretch vibration);1 064.97 cm-1(N-N stretch vibration); and 737.25 cm-1(benzene ring out-of-plane deformation).
The FT-IR spectra of BSC (KBr) are identified at:3 065.31 cm-1(O-H stretch vibration); 2 922.73 cm-1and 2 851.55 cm-1(C-H stretch vibration); 1 723.25 cm-1(C=O stretch vibration); 1 590.01 cm-1and 1 495.36 cm-1(benzene ring stretch vibration); 1 411.28 cm-1(N=N stretch vibration); 1 301.44 cm-1(arylamine C-N stretch vibration); 1 203.85 cm-1(C=S stretch vibration);1 064.83 cm-1(N-N stretch vibration); 737.67 cm-1(benzene ring out-of-plane deformation); and 669.83 cm-1(C-S stretch vibration). Particularly, due to conjugating to C=S, the spectrum at 1 565.39 cm-1(-(S=)C-N) , is found to be higher in frequency.
The results of elemental analysis are presented in Table 1. The measured values of the derivatives were in good agreement with the theoretical values. The results of FTIR spectroscopy and elemental analysis demonstrated that BC and BSC had been successfully synthesized.
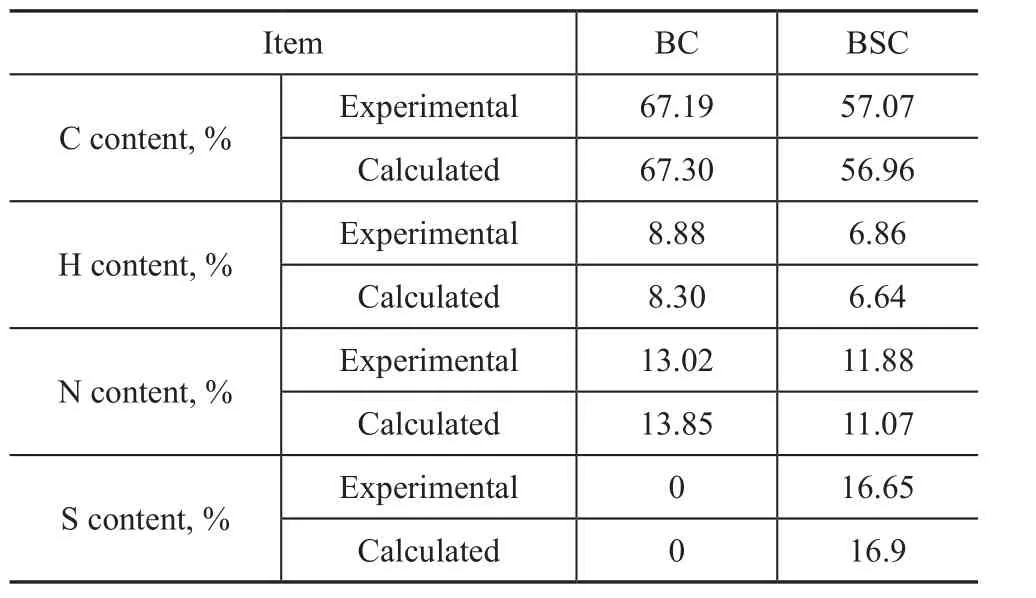
Table 1 Elemental analysis results of the synthesized benzotriazole derivatives
2.2 Friction and wear test
Prior to the test, the base fluid was prepared by incorporating 3.0% of triethanolamine into the waterglycol fluid to enhance the solubility of the as-synthesized compounds. The water-glycol fluid consisted of 42.0%of deionized water, 42.0% of diethylene glycol, and 16% of polyether with an average molecular weight of 40 000. Physiochemical indices of the base fluid are listed in Table 2. Then the fluids containing different concentrations of BC and BSC, viz. 1.0%, 2.0% and 3.0%,respectively, were prepared, as shown in Figure 2. It can be obviously seen from Figure 2 that the fluids formulated with BC and BSC were transparent, indicating that both BC and BSC possessed outstanding solubility in the base fluid.
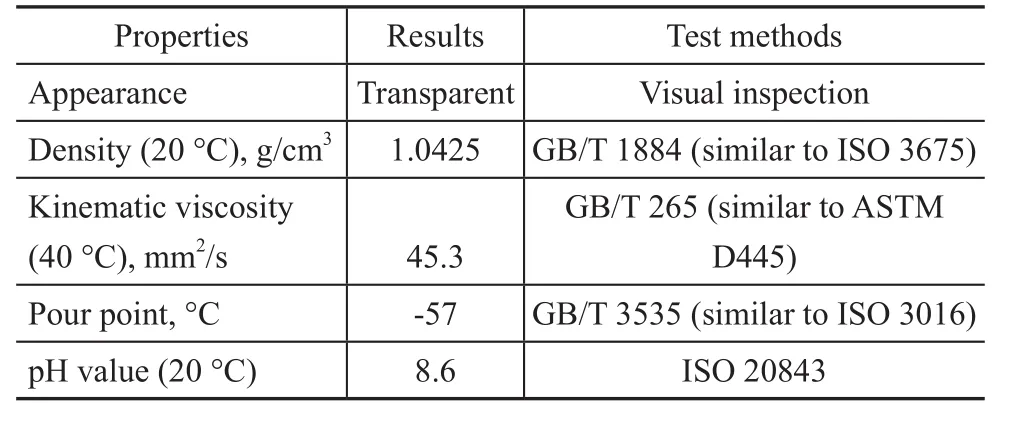
Table 2 Physiochemical properties of base fluid

Figure 2 Photographs of base fluid and fluids containing synthesized additives
The anti-wear property of BC and BSC was evaluated on a MMW-1 four ball tribotester (made by the Jinan Testing Machine Factory, China). The experiments were carried out at room temperature at a revolution rate of 1 200 r/min over a test duration of 30 min. The balls (with a diameter of 12.7 mm and a HRC of 59―61) were made of ASTM E52100 bearing steel with a surface roughness of 0.040 μm. After each test run, the wear scar diameter (WSD) of three lower balls was measured by an optical microscope to the accuracy of 0.01 mm, and the duplicated tests should be conducted if the relative error of the WSDs was larger than 10%. Then, the average of the WSDs was cited as the reported value.
The friction-reducing property of BC and BSC was measured on a MTF-3000 multifunctional friction and wear tester (made by Rtec, USA). The lower disc (made of ASTM E52100 bearing steel) slides reciprocally at an amplitude of 1 mm against a stationary ball (made of ASTM E52100 bearing steel, 12.7 mm in diameter). All the tests were conducted at a frequency of 20 Hz over 30 min at room temperature. The friction coefficient curves were recorded automatically by the system attached to the tester.The maximum non-seizure loads (PBvalue) and welding loads (PDvalue) of the as-synthesized additives were evaluated on a MRS-10B four ball tribo-tester (made by the Jinan Testing Machine Factory, China). All the tests were measured with the ASTM E52100 bearing steel ball (with a diameter of 12.7 mm and a HRC of 59―61) operating at a revolution rate of 1 450 rpm for 10 s, according to the Chinese national standard GB 3142―1982, which was similar to ASTM D 2783 method.
2.3 Copper corrosion-inhibiting property and antirust property
The copper corrosion-inhibiting property of the synthesized additives was evaluated by conducting copper corrosiveness test, according to the national standard GB/T 5095—2008, which was similar to the ASTM D130 method.The polished electrolytic copper strip was immersed in the base fluid with different concentration of additives. The fluid temperature was maintained at 50±1 °C for 3 hours.After the termination of test duration, the copper strip was cleaned and compared to the ASTM standard colorimetric plate to evaluate the corrosion degree.
The antirust property was evaluated according to the national standard GB 11143—1989, which was similar to ASTM 665―83. A polished steel bar was immersed in 300 mL of the fluid with different concentration of additives. Then,30 mL of deionized water was added into the fluid under continuous stirring at 60±1 °C for 24 hours, after that, the steel bar was rinsed with isooctane. The rust degree was estimated by observing the quantity and proportion of the rust spots and rust strip on the steel bar surface.
2.4 Surface analysis
Prior to the analysis, the lower balls obtained after anti-wear property tests were ultrasonically rinsed in anhydrous ethanol. The morphology and element composition of the worn surface were analyzed by a Zeiss EVO 18 scanning electron microscope (SEM) equipped with an energy dispersive spectrometer (EDS). The tribochemical species of typical elements on the worn surface were identified by a Thermo ESCALab-250 XPS.The exciting source was Al Kαand the reference binding energy was the contaminated carbon (C1s: 284.8 eV).
3 Results and Discussion
3.1 Anti-wear performance
Figure 3 shows the variation of WSD with the concentrations of the two benzotriazole derivatives under a load of 294 N and a rotary speed of 1200 r/min. It can be seen from Figure 3 that the WSD decreased with the addition of BC or BSC in all tested concentration ranges as compared to that of base fluid. As for the BC additive,the WSDs at first decreased and then turned down gradually with an increasing concentration of BC, and the WSD reached its smallest value at a BC concentration of 1.5%. In contrast, The WSD formed in the BSC-doped fluid dramatically reduced and reached its minimum value (0.41 mm) at a BSC concentration of 1.0%. After that minimum value, the WSD increased gradually with an increasing BSC concentration, which was probably ascribed to the effect of corrosion wear induced by excessive elemental sulfur contained in BSC. Water is an effective solvent for the sulfur-containing compounds, and it might well dissolve or wash away the formed tribo-film,making the fresh metal surface exposed and subjected to accelerated corrosion[22]. It should also be noted that, the WSDs formed in the BSC-doped fluid were smaller than those of BC at each tested concentration, confirming the better anti-wear ability of BSC.
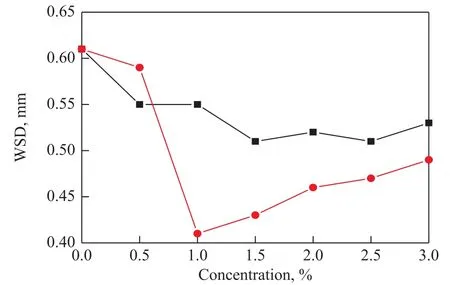
Figure 3 WSD as a function of the concentrations of BC and BSC under a load of 294 N
Figure 4 shows the variation of WSD under the investigated load for base fluid and lubricants formulated with 2.0% of BC and 2.0% of BSC, respectively. It can be seen from Figure 4 that two additives could significantly reduce the WSD of base fluid under the load ranging from 98 N to 294 N, illustrating that the assynthesized additives acted as the outstanding friction and wear reducers in the base fluid. In addition, the WSD of fluid doped with BSC was smaller than that of BC-doped fluid under each load tested. This result might be attributed to the “active elements” sulfur and nitrogen contained in BSC, which could facilitate the formation of tribochemical reaction film and adsorption film, thus exhibiting better anti-wear property under the relatively higher load as compared to that of BC.
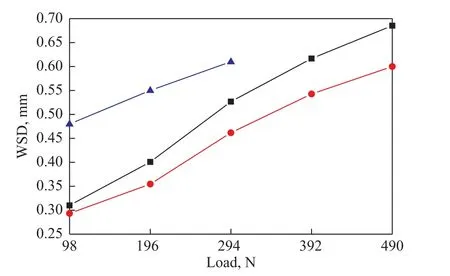
Figure 4 Variation of WSD with loads
3.2 Friction-reducing property
The variation of friction coefficients with the concentration of BC and BSC under a load of 300 N is presented in Figure 5. It can be seen from Figure 5 that the friction coefficients of both additives decreased with an increasing additive concentration, but slightly varied at the additive concentration of 0.5%. This demonstrated that both BC and BSC could efficiently reduce the friction coefficients of base fluid. Moreover, the friction coefficients of the BSC-formulated fluid were always smaller than those of BC-formulated fluid in the range of investigated concentrations (0.5%—3.0%), indicating that BSC was more effective in reducing friction coefficients than BC.
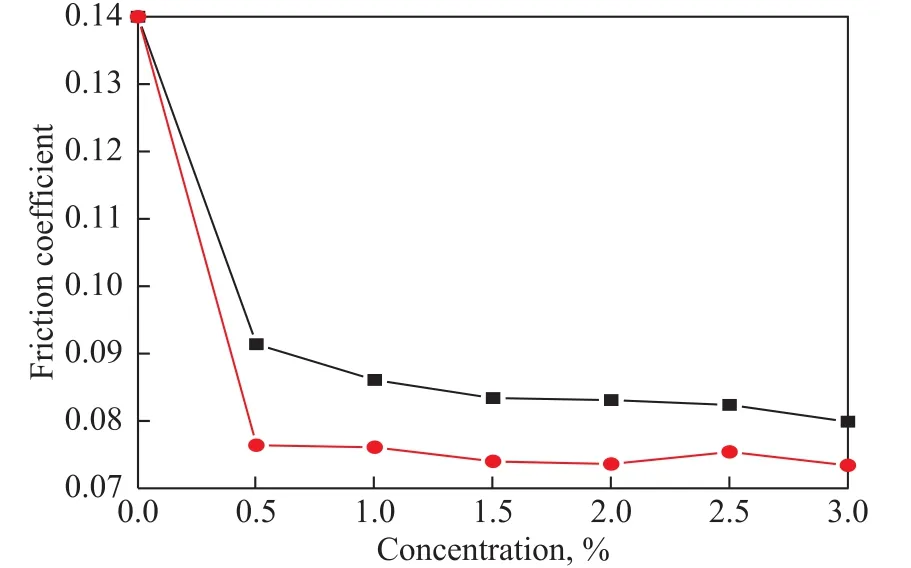
Figure 5 Friction coefficient as a function of additive concentration under a load of 300 N
Figure 6 reveals the variation of friction coefficients of base fluid and fluids containing 2.0% of BC and 2.0% of BSC, respectively, under the load investigated thereby.It is obvious from Figure 6 that the friction coefficient of the BC-doped fluid was lower than that of the BSC-doped fluid under load in the range of 100—200 N, but BSC had better friction-reducing ability than that of the BC-doped fluid under relatively high loads (300—500 N). Thus, it can be inferred that the absorption film generated by BC was the main protector in reducing friction coefficient under a relatively low load, and the tribo-chemical reaction film formed by BSC played a dominant role in lowering friction coefficients at a relatively high load.
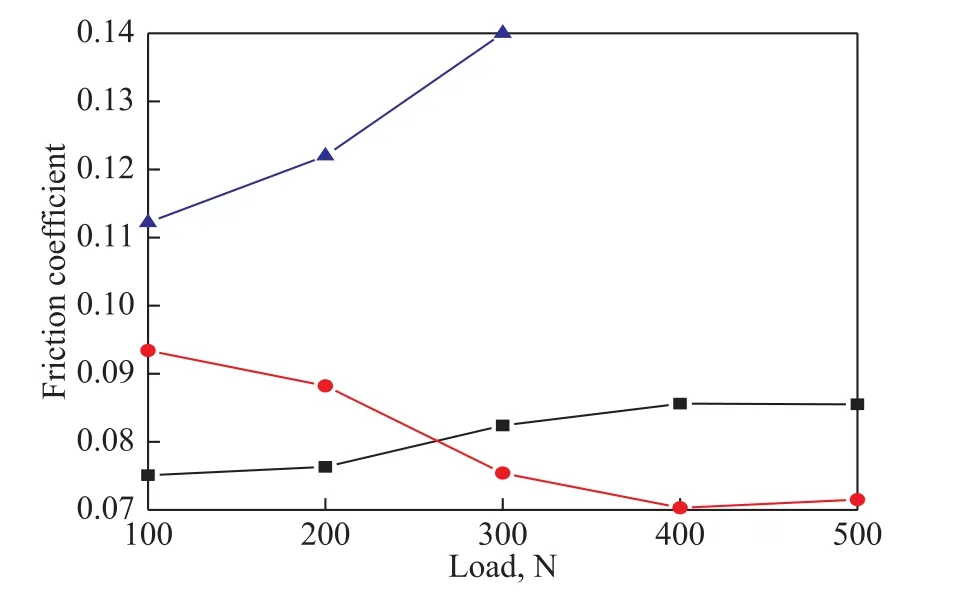
Figure 6 Friction coefficient as a function of the load at an additive concentration of 2%
3.3 Copper corrosion-inhibiting property and antirust property
The performance indicators of the anticorrosion test and antirust test are listed in Table 4. The status of anticorrosion performance was evaluated on the basis of the color of tarnished copper strip after test. According to the classification of corrosion standard colorimetric plate,the low-tarnish class denotes the better anticorrosion property. As shown in Table 4, the class of corrosiveness to copper by base fluid was 1b, while that by the fluids containing additives at all the tested concentration levels was 1a. The result indicated that both BC and BSC had improved copper corrosion-inhibiting property. This fact has verified that in an alkaline medium of the base fluid,BTA could be adsorbed on the copper surface to generate a polymer film containing the copper oxide, and therefore the strong polymer film could prevent other ions from eroding the copper surface[23].
The results of rust-inhibiting tests of BC and BSC are shown in Table 4. It can be seen that slight rusting occurred on the steel bar immersed in the base fluid. The antirust property of base fluid was further improved with the addition of BC and BSC since no rust spots had been observed, which could be attributed to the protective films generated by the triazole ring and alkylcarboxyl groups[24].

Table 4 Classification of anticorrosion and antirust performance of fluids with different additive concentrations
3.4 Load-carrying capacity
The maximum non-seizure loads (PBvalue) and welding loads (PDvalue) of the fluids containing various concentrations of the two as-prepared additives are listed in Table 3. As illustrated in Table 3, BC or BSC could significantly improve the PBvalue as compared to that of base fluid, even at a low concentration of 1%. The PBvalue of BC-doped lubricants and BCS-doped lubricants reached the highest value when the additive mass fraction was 2% and 3%, respectively. Thus, it can be deduced that elemental sulfur coming from BSC could help generate a tribological reaction film to bear the loads at a sufficient extent.
As regards the PDvalue, the BC additive could not make any improvement irrespective of its content in the base fluid, but the PDvalue of BSC increased to 1 961 N at an additive content of 2.0% in base fluid. Therefore, it can be concluded that BSC additive exhibited better ability in bearing the extreme pressure.
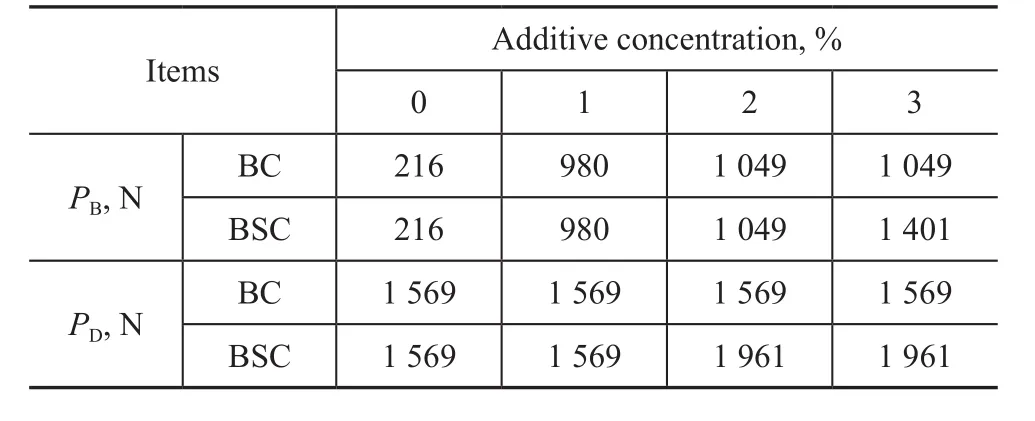
Table 3 PB and PD value of fluids with different additive concentrations
3.5 Morphology of worn surfaces
Figure 7 shows the morphologies of worn surface of steel balls lubricated by the neat fluid and the formulated lubricants under a load of 294 N and at a rotary speed of 1 200 r/min for 30 min. The morphologies on the left side are overall perspectives of the worn surfaces, whereas the right ones are the details. It could be seen from Figure 7 that:Surface of the steel ball lubricated by base fluid exhibited many deep grooves with a significant amount of pits and spalls identified, indicating that obvious abrasive wear and adhesive wear occurred on the rubbing surface.
The wear scar of steel balls lubricated with the BC-doped fluids (Figure 7c,7d, 7e, 7f) and the BSC-doped fluids(Figure 7g, 7h, 7i,7j) were smaller and smoother than those lubricated with the base fluid, indicating that both BC and BSC could effectively reduce friction and wear;The polishing scratch of steel ball lubricated by the BSC-doped lubricants (Figure 7g, 7h, 7i, 7j) was slighter than that that lubricated by the BC-doped lubricants (Figure 7c, 7d, 7e, 7f), attesting to the more desirable effect of BSC on reducing friction and wear;
The wear scar diameter of steel ball lubricated with the fluid containing 3.0% of BSC (Figure 7i) was a little bigger than that obtained from the fluid containing 1.0%of BSC (Figure 7g), indicating an excessive incorporation of BSC was not desirable in improving anti-wear performance. This outcome might be attributed to the corrosion wear induced by water and excess elemental sulfur in the additive[22]. The previous analysis agreed well with the results of tribological test shown in Figure 5.

Figure 7 SEM morphologies of worn surface of steel balls lubricated by different samples under a load of 294 N
3.6 Element composition
Table 5 lists the concentration of elements on the worn surface of steel balls lubricated with base fluids or formulated lubricants after four-ball tests under a load of 294 N by EDS analysis. It can be seen from Table 5 that the concentrations of elemental carbon and oxygen on the worn surface lubricated with BC or BSC-containing fluids were lower as compared to that obtained from the base fluid, and elemental nitrogen and sulfur could be detected on the worn surface lubricated by the formulated fluids, indicating that the nitrogen or sulfur-containing compounds were generated in the course of friction.
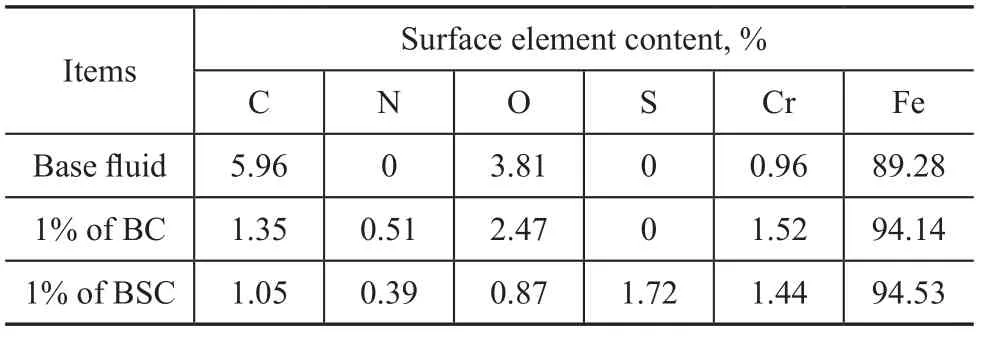
Table 5 Surface element content of new steel ball and worn surface tested by EDS
3.7 Tribochemical characteristics
The XPS spectra of typical elements on the worn surface of steel balls lubricated by 2.0% of BC and 2.0% of BSC are showed in Figure 8 and Figure 9, respectively.
In Figure 8, the peak of C1s (Figure 8a) at a binding energy of 284.7 eV was attributed to the C-C bond,indicating that the lubricant molecules were adsorbed on the metal surface. The binding energy of Fe2p at 710.5 eV and 724.1 eV (Figure 8b) belonged to Fe2O3or FeOOH,respectively[25], which was consistent with the peak of O 1s at a binding energy of around 531.7 eV (Figure 8c).In the spectrum of N1s, the peak at a binding energy of 399.8 eV (Figure 8d), had illustrated that the nitrogencontaining organics were formed on the metal surface during the sliding process. Thus, it can be inferred that a composite boundary lubrication film mainly composed of Fe2O3, FeOOH and the nitrogen-containing compounds was formed on the surface.
In Figure 9, the binding energy of Fe2p at 710.7 eV,724.1 eV, and 713.2 eV (Figure 9b) was corresponding to Fe2O3, FeOOH or FeS, and FeSO4[26], respectively, in combination with the peak of O1s around the binding energy of 532.1 eV (Figure 9c), as well as the peaks of S2p located at 160.1 eV and 168.1 eV (Figure 9d). In the spectrum of N1s (Figure 9e), the peak at the binding energy of 399.6 eV indicated that the nitrogen-containing compounds were adsorbed on the metal surface. The XPS results in Figure 9 demonstrated that the complicated tribo-chemical reaction film on the surface mainly consisted Fe2O3, FeOOH or FeS, and FeSO4, as well as the nitrogen-containing organics.
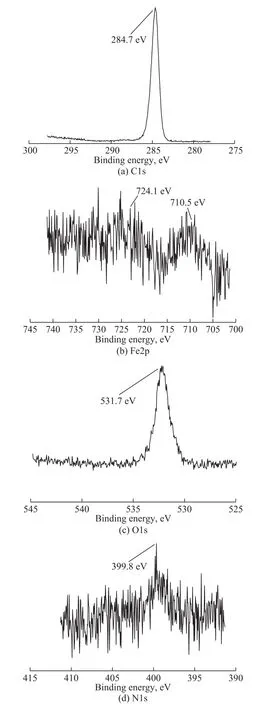
Figure 8 XPS spectra of typical elements on the worn surfaces lubricated with 2% of BC after four-ball test conducted under a load of 294 N for 30 minutes
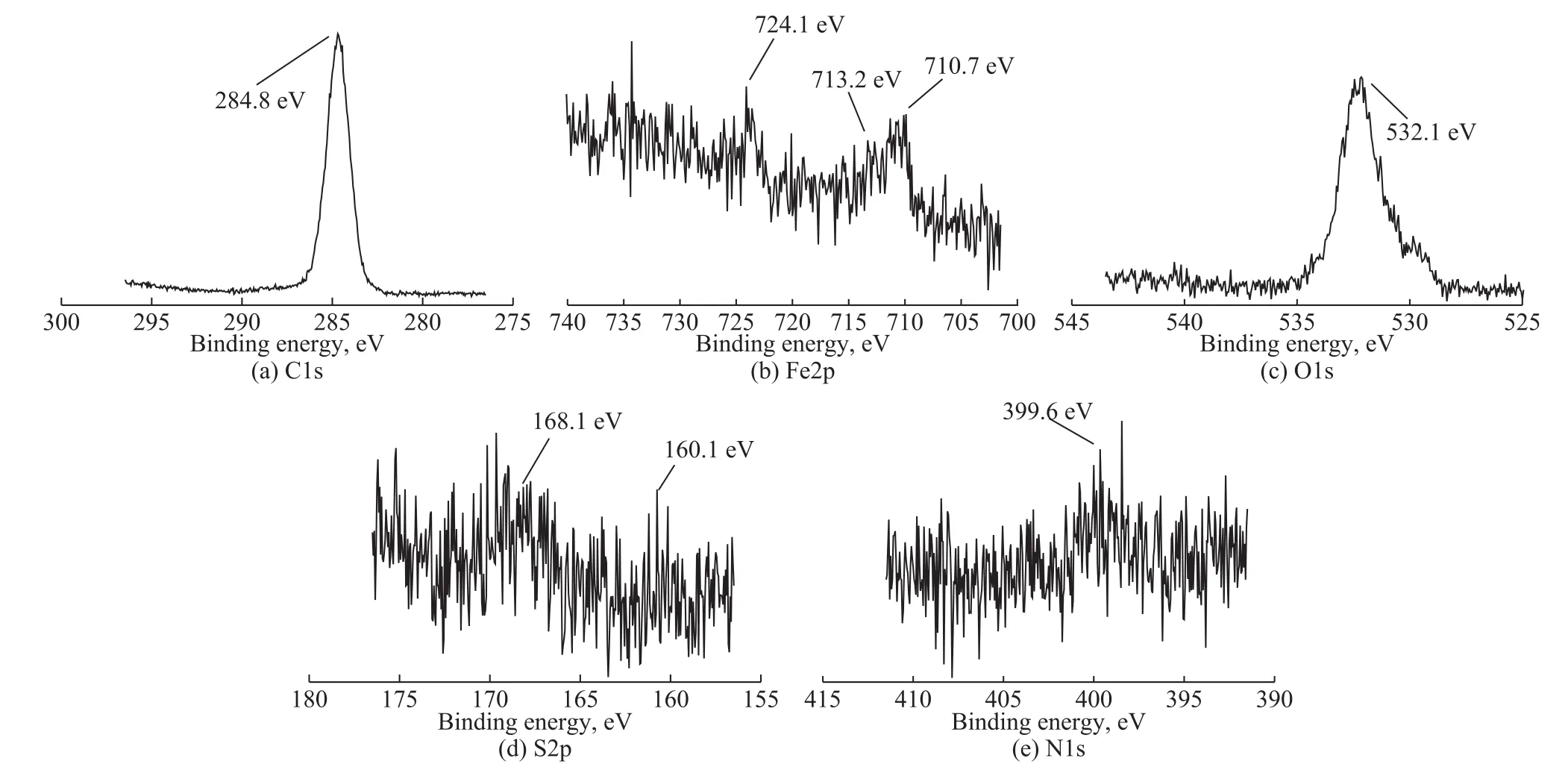
Figure 9 XPS spectra of typical elements on the worn surfaces lubricated with 2% of BSC after four-ball test conducted under a load of 294 N for 30 minutes.
Judging from the results of analysis mentioned above,it could be concluded that BC and BSC had different lubricating mechanisms. The additive BC could be adsorbed on the metal surface to generate the nitrogencontaining protective films. Compared with BC, under a high load of the lubricating conditions, the sulfurcontaining BSC could form ferrous sulfide and iron sulfates film which had better lubricity than the absorption film generated by BC. Nevertheless, it should be noted that the excess BSC could induce wear corrosion in the water-based lubrication.
According to the previous analysis, we deduced the tribological mechanism of BC and BSC as follows: In the initial stage, both BC and BSC could steadily be adsorbed on the metal surface because of their terminal carboxyl groups,and a rigid straight chain of about 10 carbon atoms could provide them with enough strength to prevent the asperity on the rubbing surface from directly contacting when a relatively low load was applied. With the increase of loads,the tribo-chemical reaction would occur to form chemical reaction films to resist friction and wear. As a consequence,both the adsorption film and the tribo-chemical reaction film should be responsible for facilitating the tribological performance of water-glycol hydraulic fluid.
4 Conclusions
1) The as-synthesized two benzotriazole derivatives, BC and BSC used as additives, could effectively facilitate the anti-wear property and friction-reducing performance of the water-glycol base fluid. The additives BC and BSC could significantly improve the PBvalues of base fluid.BC could not increase the PDvalues irrespective of a range of concentrations adopted thereby, but BSC could obviously enhance the PDvalue when its concentration was greater than 2.0%.
2) Both BC and BSC had excellent copper corrosioninhibiting performance and antirust property, which obviously would determine their potential application value.
3) The worn surface analysis illustrated that both the adsorption film and the tribo-chemical films on the surfaces lubricated with formulated lubricants were mainly composed of iron oxides, nitrogen-containing compounds as well as iron sulfides and iron sulfates,which should be responsible for the facilitated anti-wear and friction-reducing performance.
4) The analysis results demonstrated the effectiveness of elemental sulfur contained in BSC in reducing the friction and wear. However, a relatively high content of elemental sulfur would have a negative impact on the anti-wear property because of the corrosion wear that could occur in the water-based fluids. As a result, concerns should be paid over selecting the proper additive according to the working conditions.
Acknowledgement:This study was financially supported by the National Natural Science Foundation of China(Grant No.51375491), the Basic Science and Frontier Technology Research Project of Chongqing (Grant No. CSTC,2017jcyjAX0058), and the Young Foundation of the Logistical Engineering University (Grant No.YQ16-420801).
- 中國(guó)煉油與石油化工的其它文章
- Effect of Biodiesel Soot on Tribological Behavior of Liquid Paraffin
- Heat Transfer Investigation and Modeling of Heat Integrated Distillation Column
- Novel Control Structure Design of Differential Pressure Thermally Coupled Reactive Distillation for Methyl Acetate Hydrolysis
- Preparation of Sodium Cobalt Tetracarbonyl and Optimization of Process Conditions for Hydroesterification of Ethylene Oxide
- Bulk Ni-Mo Composites Prepared by Solid Reaction Method and Their Hydrodeoxygenation Performance
- Study on Synthesis of Mesoporous M-MCM-48 (M = Zr, Mg)and Its Activity for Isomerization of n-Heptane

