Study on Synthesis of Mesoporous M-MCM-48 (M = Zr, Mg)and Its Activity for Isomerization of n-Heptane
Wang Yingjun; Dong Xiuli; Ma Shoutao; Wang Jian; Li Xiaoqing; Suo Yanhua; Zhang Wei
(College of Chemistry and Chemical Engineering, Northeast Petroleum University, Daqing 163318)
Abstract: The mesoporous materials supported zirconium (Zr-MCM-48) and magnesium (Mg-MCM-48) with three-dimensional pore structures and different Zr or Mg contents were prepared via the hydrothermal method, respectively. The prepared samples were characterized by means of X-ray diffraction (XRD), scanning electron microscope (SEM), transmission electron microscope (TEM), Fourier transform infrared spectroscopy (FT-IR), NH3 temperature programmed desorption(NH3-TPD) and N2 adsorption-desorption. The XRD, TEM and N2 adsorption-desorption results suggested that M-MCM-48(where M = Zr, Mg) samples still maintained typical cubic mesoporous framework of MCM-48, with slight decrease of specific surface areas and mesopore orders. The isomerization of n-heptane was carried out as a probe reaction at various factors including the zirconium or magnesium content, reaction time, reaction temperature and weight hourly space velocity (WHSV). Preliminary results demonstrated that the Mg-MCM-48 catalyst exhibited a higher catalytic activity with a maximum heptane conversion of 91.5% and a corresponding isomerization selectivity of 72.3% as compared to that of Zr-MCM-48. However, the catalytic stability of Mg-MCM-48 for n-heptane isomerization was poorer than Zr-MCM-48, because the Mg-MCM-48 catalyst could maintain a relatively high catalytic activity for only 130 min, while the Zr-MCM-48 catalyst could maintain a relatively high catalytic activity for more than 190 min without any obvious decrease in performance.
Key words: n-heptane; hydroisomerization; MCM-48; zirconium; magnesium
1 Introduction
Isomerization of alkanes or alkenes plays an important role in the manufacture of high quality automotive fuel because it can increase the octane number. With the increasing emphasis on environment and the continuous growth on demand for clean fuel, the production of environmentally friendly high-octane gasoline has received widespread attention[1]. It is well known that consecutive branching reactions over bifunctional metal-acid catalysts are applicable to alkane isomerization process. The acidity of bifunctional catalysts can be supplied by zeolites and the dehydrogenation/hydrogenation function is usually provided by noble metals like palladium, platinum, and ruthenium. Typically, owing to the versatile application of bifunctional metal/acid catalysts, especially in isomerization of n-C5/C6-alkanes, a lot of researches have been carried out in this area.
MCM-48 is an ideal support material because it possesses high surface area, large pore volume and single pore distribution which can allow larger molecules or groups to enter their pores. Besides, MCM-48 has a three-dimensional cubic Ia3D mesostructure consisting of two interpenetrating continuous networks of chiral channels.This unique 3D channel network is judged to supply a highly opened porous host in the catalytic reactions which can facilitate the inclusion or diffusion of the materials throughout the pore channels without pore blockage[2-3].But pure silicon MCM-48 lacks weak acidity sites which can lead to low catalytic activity for isomerization of alkanes, so synthesis of MCM-48 with high acidity and catalytic activity is the pursued goal of researchers. The surface acidity of mesoporous materials can be effectively enhanced through the multifunctionalization approach. It is reported that the surface Br?nsted acidity can be generated and the total number of acid sites are noticeably increased by grafting some strong acid sites like sulfate ions[4]or heteropolyacids[5]onto the surface of mesoporous material MCM-48. Also, when the trivalent metal ions(M3+) are doped into the zeolite framework, the M3+ions embraced by Si4+ions can lead to the charge imbalance,which is prone to generating strong Lewis acid or Br?nsted acid centers[6]. Until now, many efforts have been made on the incorporation of various heteroatoms such as Sn[7],Cr[8], and Mn[9]into the framework of the pure silica mesoporous molecular sieves. The catalytic activity of some mesoporous catalysts had been evaluated via the tertiary butylation of phenol[10]or ethene to propene reaction[11],but little attention has been paid to the investigation of alkanes isomerization.
For this purpose, the Zr-MCM-48 and Mg-MCM-48 mesoporous materials were synthesized by the hydrothermal method based on the self-assembly theory in the present work. Particularly, the effects of zirconium or magnesium content, WHSV, the reaction time, and the reaction temperature on the n-heptane conversion and isomerization selectivity were investigated.
2 Experimental
2.1 Materials
All chemicals used in this work, such as tetraethyl orthosilicate (TEOS), zirconium nitrate (Zr(NO)4·5H2O),cetyltrimethyl ammonium bromide (CTAB), sodium hydroxide (NaOH), NaF, n-heptane, and deionized water were purchased from the Tianjin Chemical Reagent Corporation, China.
2.2 Catalyst preparation
MCM-48 was prepared by the hydrothermal method using cetyltrimethylammonium bromide (CTAB) as the structure directing agent, and tetraethyl orthosilicate (TEOS) as the silicon source at a molar ratio consisting of 1 TEOS: 0.65 CTAB: 0.5 NaOH: 62 H2O. As for M-MCM-48 (where M = Zr, Mg), the samples were synthesized by the hydrothermal method along with the gel composition consisting of 1 TEOS: x Mg (or y Zr): 0.65 CTAB: 0.49 NaOH: 62 H2O (where x = 0.01, 0.02, 0.03, 0.04 or y = 0.01, 0.02,0.03, 0.04…0.1), and the typical procedure was conducted as follows: A given amount of Zr(NO)4·5H2O, 0.45 g of NaOH and 0.09 g of NaF were dissolved in 25 mL of distilled water, and then 5.3 g of CTAB were added into the mixture under vigorous stirring for 30 min at 35 °C. After that, 5 mL of TEOS were slowly added into the above mixture. After stirring for another 60 min, the resulting mixture was transferred into a 50-mL Teflon-lined stainless autoclave and was subject to crystallization at 110 °C for 24 h in an oven. The obtained solid product was filtered, washed with deionized water, and dried at 60 °C for 24 h. The dried powder was then ground finely and calcined at 550 °C for 6 h in air at a heating rate of 2 °C/min. The calcined sample was denoted as Zr-MCM-48. The procedure for synthesis of Mg-MCM-48 was similar to that of Zr-MCM-48, and the synthesized samples were labeled as M-MCM-48-x,where M represents Mg or Zr, and x is the molar ratio of Si/M in the synthesized gel.
2.3 Catalyst characterization
The powder XRD patterns were recorded on a Rigaku D/max-IIB diffractometer using Cu Kα radiation. The scanning speed was 2(°)/min and the scanning range was 1.5°―10°. The N2adsorption-desorption experiments were conducted with an American Quantachrome NOVA/2000e surface area and pore size analyzer, using liquid nitrogen as the adsorbate. The FT-IR spectra were recorded on a Thermo Nicolet AVATAR-360 with the KBr pellet technique. The FT-IR spectrometer operated in the range of 450―4 000 cm-1. The scanning electron microscopy (SEM) images were taken on a ZEISS SUPRA 55 SAPPHIRE operating at an acceleration voltage of 20 kV.The TEM images were recorded on a TECNAI G2F20STWIN transmission electron microscope operating at an acceleration voltage of 100―120 kV. The acidic properties of samples were tested via NH3-TPD with an Autochem II 2920 chemisorption analyzer, which used 0.15 g of samples, the temperature of which was increased to 450 °C under N2flow (at a rate of 40 mL/min) and was then kept at that value for 2 h, and afterwards was gradually reduced to 150 °C. The samples adsorbed ammonia to saturation at 150 °C for 0.5 h. The ammonia adsorbed on the surface of the sample was purged with helium and then the temperature was raised to 700 °C, at which the TCD could detect the desorption signal.
2.4 Catalytic activity test
The n-heptane hydroisomerization was performed by a self-assembled continuous flow fixed-bed stainless steel reactor, in which the reaction was carried out under normal pressure. A total of 0.2 g of the catalyst mixed with the quartz sand was placed in the reactor. The mixture was at first subjected to reduction at 673 K for 3 h. Then the catalytic reaction was conducted under the same conditions with the reaction gas (H2/n-C7) introduced at a suitable reaction temperature. After the reaction was stabilized for 30 min, the products were analyzed by a GC-7980A type gas chromatograph.
3 Results and Discussion
3.1 Catalyst characterization
3.1.1 XRD analysis
Figure 1 shows the XRD patterns of various samples. It can be seen from Figure 1a that when the metal content was not high (at Mg/Si molar ratio ≤0.03), the characteristic diffraction peaks (211) and (220) of the samples could be observed, suggesting that the incorporation of Mg in the silica matrix did not destroy the long-range ordered structure of MCM-48. When the molar ratio of Mg/Si was 0.04, the diffraction peaks of MCM-48 could not be observed, which could be attributed to the excessive amount of the doping metal that destroyed the condition for formation of MCM-48. As for Zr-MCM-48 (Figure 1b), its diffraction peaks could be observed obviously when the molar ratio of Zr/Si was 0.1, which implied that the sustainable amount of doped Zr metal was more than that of Mg in MCM-48. The above phenomenon could occur probably because the charges of Zr4+was the same as that of the framework Si4+, and the charges of Zr4+and Si4+could match better during synthesis of the mesoporous Zr-MCM-48, which might lead to a higher sustainable doping amount of Zr than that of Mg. As the charges of Mg2+were fewer than that of the framework Si4+, then the charges of Mg2+and Si4+would match poorer during synthesis of the mesoporous Mg-MCM-48, which might lead to a lower sustainable amount of doped Mg than that of Zr. Therefore, the characteristic diffraction peaks (211)and (220) of all the Zr-MCM-48 samples (with Zr/Si molar ratio = 0.02―0.1) could be observed, and when the molar ratio of Mg/Si was 0.04, the MCM-48 diffraction peaks could not be observed.
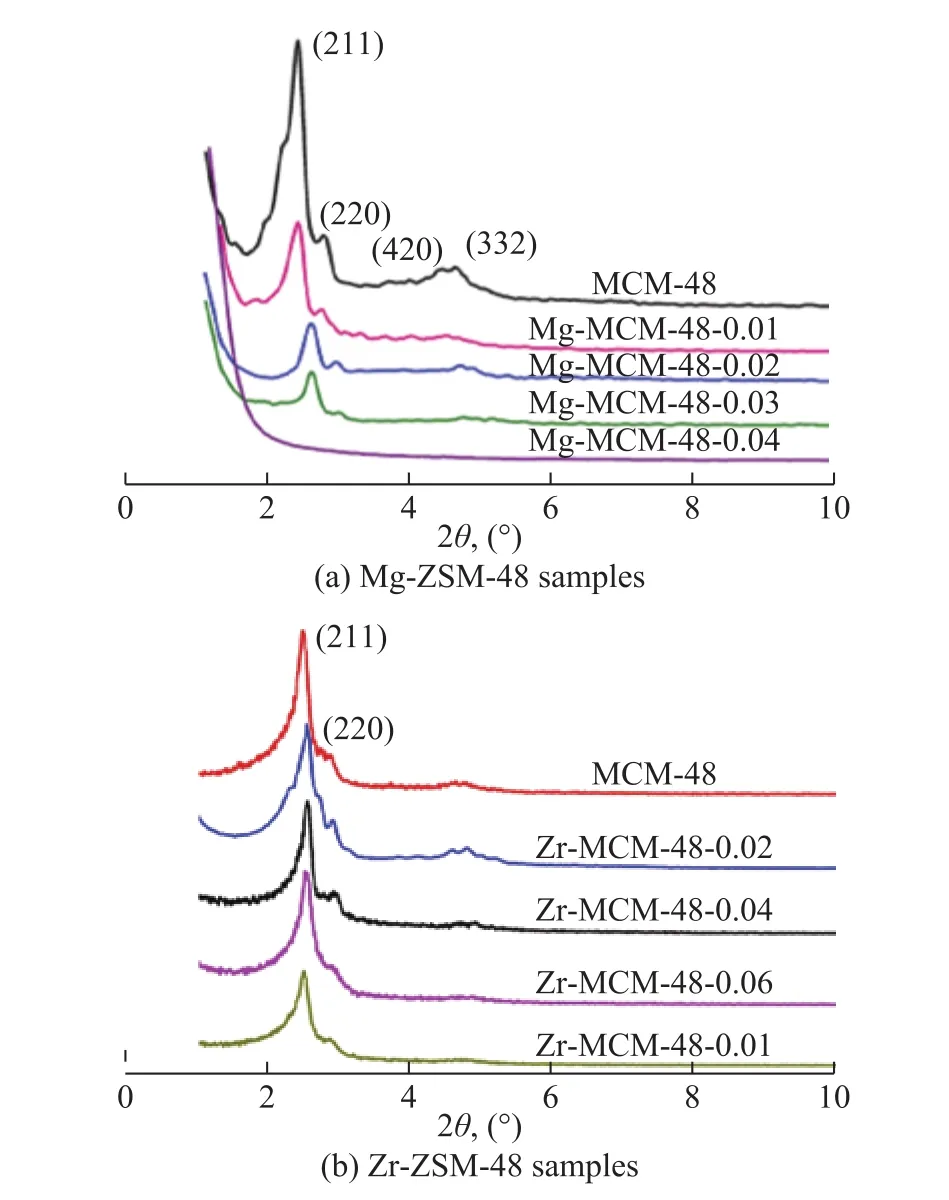
Figure 1 X-ray diffraction patterns of the prepared Mg-MCM-48 samples and Zr-MCM-48 samples
3.1.2 NH3-TPD analysis
The distribution of acid sites in M-MCM-48 samples was determined by NH3temperature-programmed desorption,with the profiles shown in Figure 2. It is well-known that the desorption peak at low temperature is related to the weak acid sites, while the desorption peak at high temperature denotes the strong acid sites, and the peak area is related to the total acid amount[12]. According to the literature[13], the desorption peak around 150―170 °C is assigned to the surface hydroxyl groups. The two desorption peaks in the range of 180―210 °C and 270―330 °C originate from the moderate and the strong acid sites,respectively, which are caused by the presence of bivalent magnesium (or other metal) in two different framework positions. The acidity of M-MCM-48 depends on the amount of Mg or Zr in the sample. In the NH3-TPD profiles of Mg-MCM-48, a main desorption peak at 220 °C can be observed, and the peak near 220 °C is produced by the weak bonding of ammonia with the samples[14].The weak desorption peak near 300 °C arises from the presence of magnesium, which is located in two different framework positions and its intensity increases with an increasing Mg content, which indicates that doping metal can improve the acidity of mesoporous molecular sieves.As regards the Zr-MCM-48 samples, one desorption peak around 180 °C can be obviously observed, illustrating that Zr-MCM-48 samples have a certain amount of medium strong acid sites, while the strong acid sites are lacking. Therefore, it is viable to make the number of the medium strong acid sites increase with the increasing Zr content.
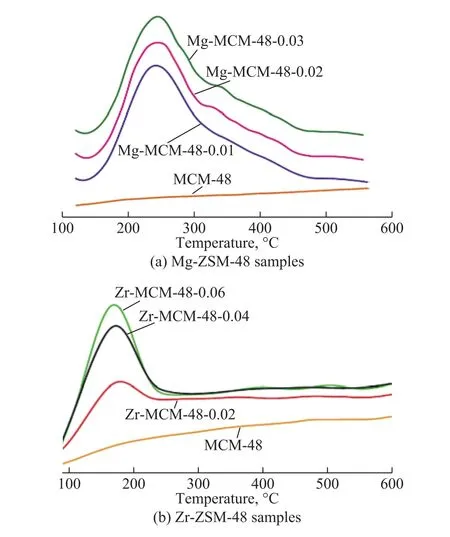
Figure 2 NH3-TPD patterns of the MCM-48 samples with different Mg contents (a) and Zr contents (b)
3.1.3 Results of physical adsorption of nitrogen
The textural properties of MCM-48, Mg-MCM-48-x(x=0.01, 0.02, 0.03) and Zr-MCM-48-x (x=0.02, 0.04,0.06) are listed in Table 1. In comparison with MCM-48,the specific surface area and pore volume of all samples decreased with an increasing content of incorporated metal (Mg or Zr). This could be probably attributed to the loss of the lattice ordering of MCM-48 framework resulted from the increase in the content of incorporated metal. The degree for loss of the lattice ordering of MCM-48 framework would become greater with an increasing incorporated metal content, and thus the specific surface area and pore volume of the samples could become smaller with the increase in the incorporated metal content.

Table 1 Textural properties of MCM-48, Mg-MCM-48-x and Zr-MCM-48-x samples
Figure 3 shows the N2adsorption-desorption isotherms of the calcined MCM-48 and M-MCM-48 samples at an M/Si molar ratio of 0.02. It is clear that the isotherms in all cases exhibit the typical Langmuir IV type adsorption isotherms with hysteresis loop, indicating that these samples have obviously typical mesoporous framework[15].Three differently well-defined stages in all isotherms can be found. At a lower relative pressure (p/p0<0.2), the adsorptive capacity increases with an increasing partial pressure, and the adsorption is caused by the adsorption of N2on the mesopore wall. When p/p0is in the range of 0.25―0.4, the curve shows a dramatic jump obviously.It can be attributed to the capillary condensation in the mesoporous channels, resulting in a rapid increase of the adsorptive capacity. Moreover the characteristics of the inflection point are presented. The sharpness and height of jumps of the curve illustrate the uniformity of the pore size[16]. This fact shows that the material has a regular form of mesoporous structure, which is consistent with the results of XRD analysis[17].
3.1.4 SEM
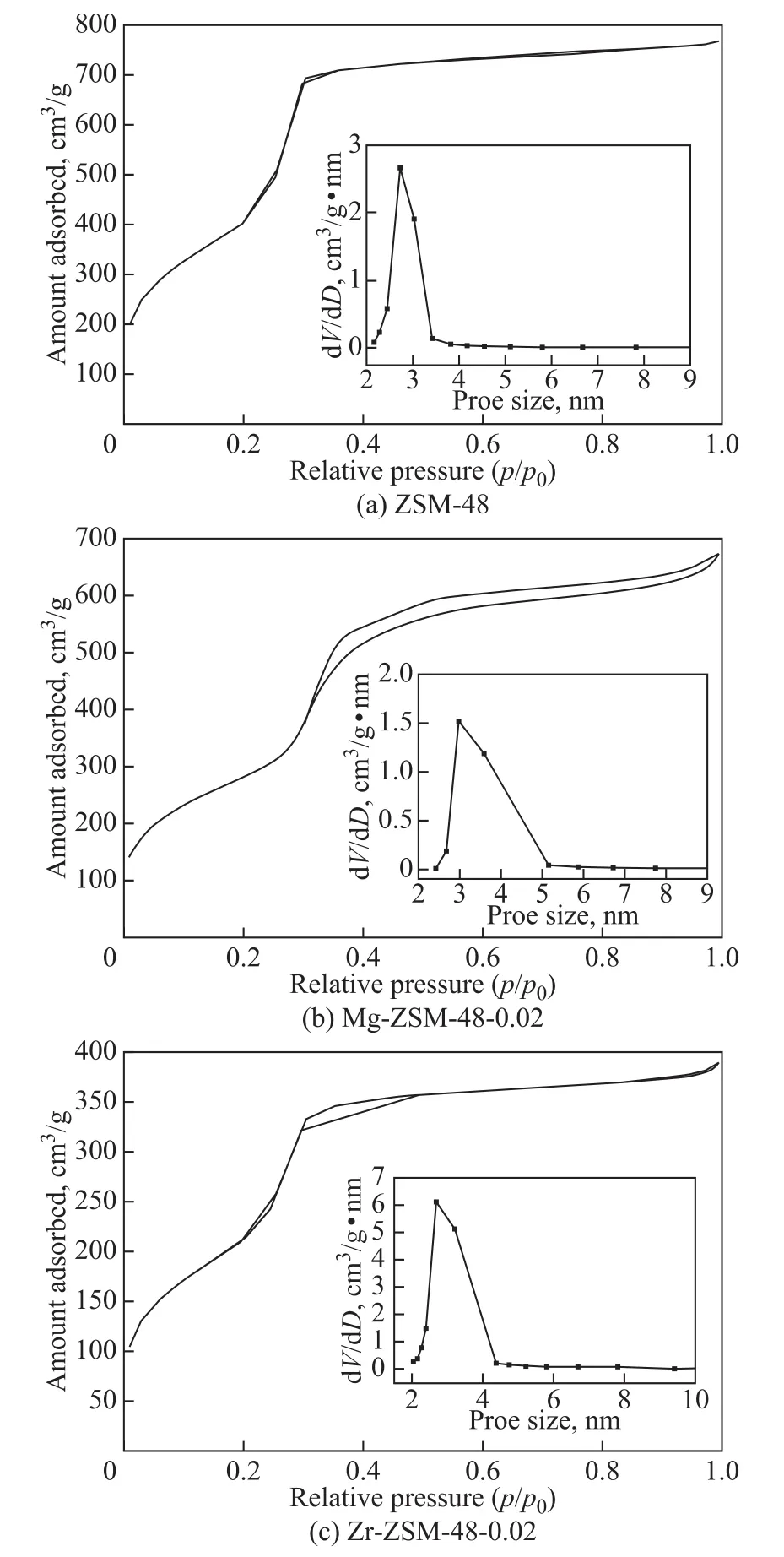
Figure 3 N2 adsorption-desorption isotherms and pore size distribution of MCM-48, Mg-MCM-48-0.02, and Zr-MCM-48-0.02
Figure 4 presents the SEM images of M-MCM-48 catalyst at an M/Si molar ratio of 0.02. The morphology of M-MCM-48 samples is almost the same as that of MCM-48. Apparently, the surface of MCM-48 sample is spherical as seen in the SEM image. However, the surface of the molecular sieve is destroyed slightly due to the high content of metal, and some sintered substances can be found on its surface. As for Zr-MCM-48, it can be seen from Figure 4 that the incorporation of Zr results in few changes in the morphology of the molecular sieve than that of Mg-MCM-48. Simultaneously, it can be seen that the particle size of Zr-MCM-48 ranges from 0.5 μm to 1 μm, while the particle size of Mg-MCM-48 is in the range of 0.3―0.5 μm, which is much smaller than that of Zr-MCM-48 and MCM-48. The reason for this phenomenon is caused by the different ion charges of Mg2+and Zr4+. The charges of Zr4+are the same as those of the framework Si4+, while the charges of Mg2+are fewer than those of the framework Si4+, which can cause the shrinkage of the framework of MCM-48, that may finally lead to the smaller particle size of Mg-MCM-48-0.02. These results are consistent with the observation of the low-angle XRD analysis and N2adsorption-desorption results. It can be inferred that Mg has a greater impact on the structure of MCM-48 than Zr.
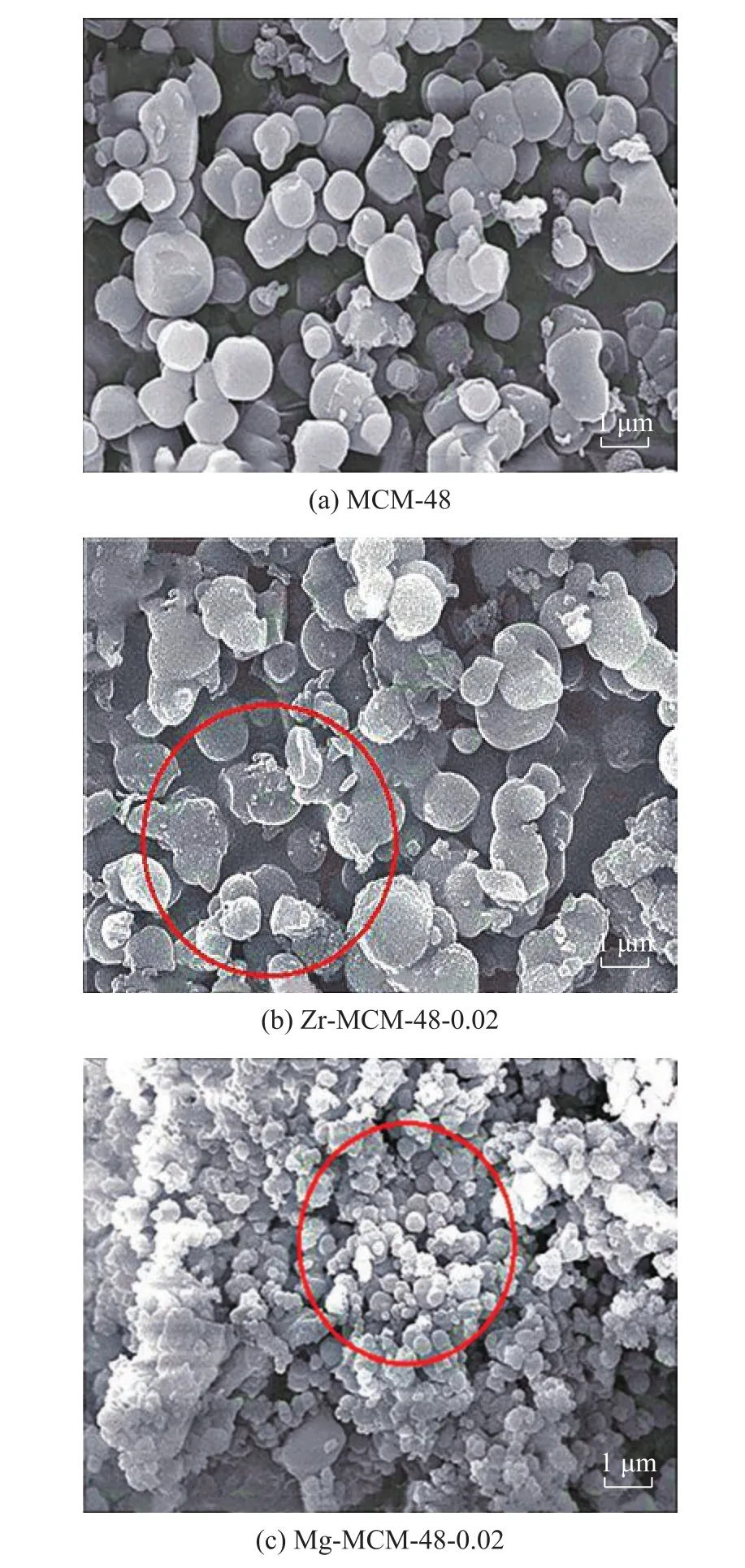
Figure 4 SEM images of the samples
3.1.5 TEM
The TEM images of the calcined MCM-48 and M-MCM-48-0.02 samples are shown in Figure 5. The pore structure of the sample is relatively complete,indicating that the Zr-MCM-48 or Mg-MCM-48 samples have the ordered mesoporous structure. And the welldefined edges can also be observed clearly, indicating that the incorporation of Mg or Zr does not damage the silica framework of MCM-48.

Figure 5 TEM images of the samples
3.1.6 Infrared spectrometry characterization (FT-IR)
Figure 6 shows the FT-IR spectra of pure silica molecular sieves Mg-MCM-48 and Zr-MCM-48 with different Zr or Mg contents. It is well known that the absorption band at 1 080 cm-1is attributed to anti-symmetric stretching vibration of the silica framework. The strong peak at 3 600 cm-1in the pure silica molecular sieve is attributed to the peak of the terminal silicon hydroxyl group attached to the surface of the mesoporous silica material.The band at 810 cm-1is related with the vibration of Si-OSi bond. The absorption peak at 960 cm-1that is found in the pure silica MCM-48 sample is usually assigned to the stretching vibrations of Si-O in the Si-O--R+groups, which is related to the v(Si-OH) or v(Si-O-Zr) vibration[18]. As for the M-MCM-48 samples, the peak intensity of 1 080 cm-1becomes stronger with an increasing zirconium or magnesium content. This phenomenon is usually regarded as an evidence for the incorporation of metal atoms in the silica framework[19]. In addition, the infrared spectrum of the antisymmetric stretching vibration of the silicon-oxygen tetrahedron is slightly shifted to the lower wave number direction. This is caused by the force constant of the Si-O bond and the Zr-O or Mg-O bond. Because the bond force constant of Zr-O or Mg-O is smaller than that of the Si-O,different bond force constant makes vibratory frequency of the Zr-O or Mg-O bond lower than the vibratory frequency of the Si-O bond, which would lead to the shift of the infrared band towards the lower frequency region.
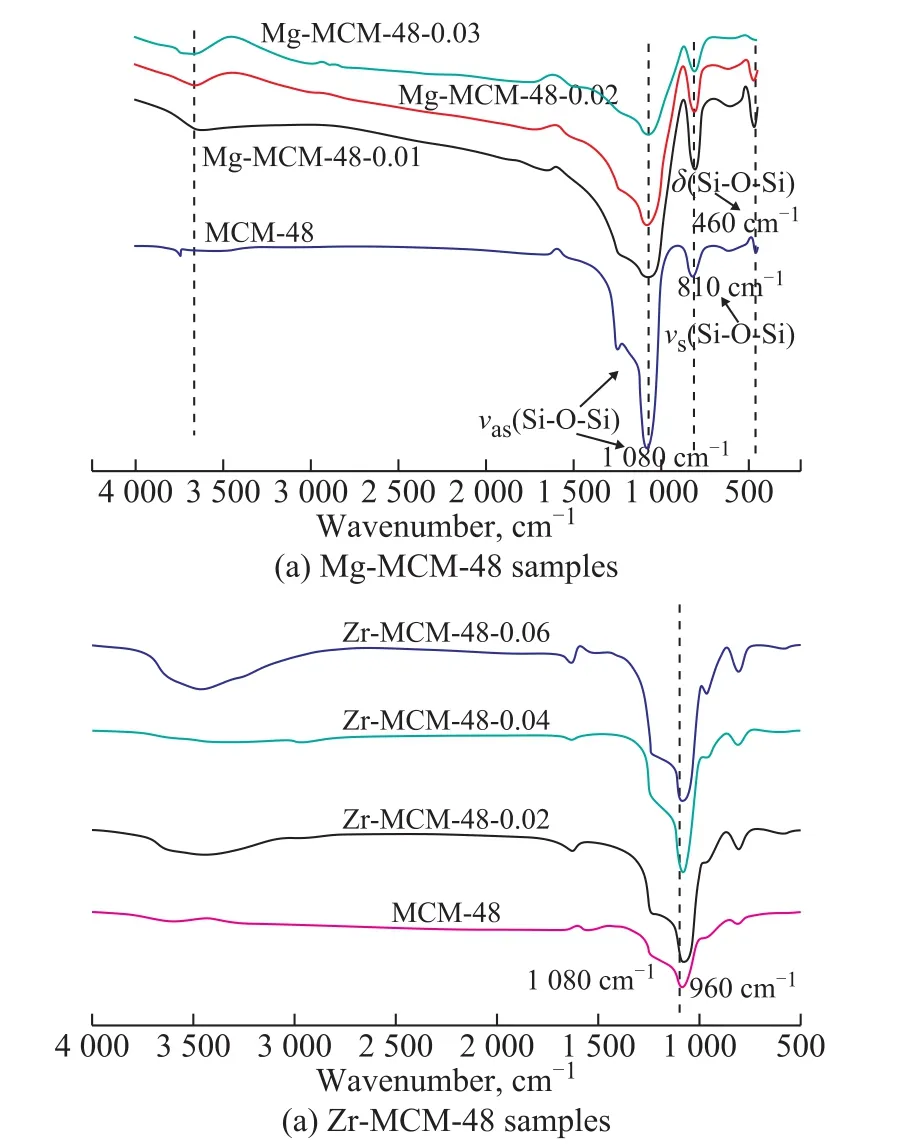
Figure 6 FT-IR spectra of Mg-MCM-48 and Zr-MCM-48 samples
3.2 Catalytic performance of different catalysts for n-heptane isomerization
3.2.1 Effect of reaction temperature and molar ratio of M/Si
Figure 7 shows the influence of Mg/Si and Zr/Si molar ratio on the catalytic performance of M-MCM-48. It can be observed that both the conversion of n-heptane and the selectivity of iso-heptane over Mg-MCM-48 increased at first and then decreased with an increasing Mg/Si molar ratio. When the Mg/Si molar ratio was 0.02, the n-heptane conversion and isoheptane selectivity reached 81.9% and 94.8%, respectively. As for Zr-MCM-48, the trends for the conversion of n-heptane and the selectivity of isoheptane were similar and the optimal molar ratio of Zr/Si was 0.03.Upon combining the NH3-TPD results of Figure 2, it can be concluded that appropriate amounts of weak acid and medium-strong acid sites are favorable to the conversion of n-heptane and the selectivity of isoheptane.
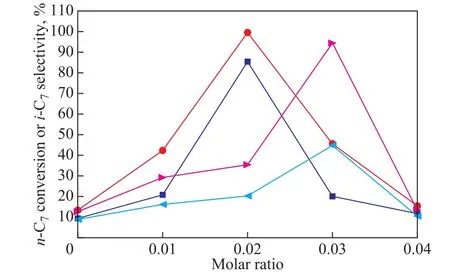
Figure 7 Influence of Mg/Si and Zr/Si molar ratio on the catalytic performance of M-MCM-48 (0.2 g of catalyst, reduction temperature = 400 °C, reduction time = 3 h, reaction temperature = 260 °C, and WHSV = 6.8 h-1)
Figure 8 shows the effect of reaction temperature on the catalytic performance of M-MCM-48 for the isomerization of n-heptane. Obviously, as the temperature varied from 220 °C to 260 °C, the n-heptane conversion on Zr-MCM-48 increased from 14.7% to 55.2%, and then decreased to 37% at 290 °C. So the optimal reaction temperature for Zr-MCM-48 was 260 °C. As for Mg-MCM-48, the n-heptane conversion increased from 15%to 92.2% when the temperature rose from 220 °C to 280 °C, and then decreased to 85% at 290 °C. Therefore the optimal reaction temperature for Mg-MCM-48 was 280 °C. One reason for the decrease of the catalytic activity with an increasing reaction temperature was probably due to the fact that the increase in reaction temperature could quicken the reaction rate, but higher reaction temperature could accelerate the carbonation of the catalyst,resulting in the diminution of the n-heptane conversion and shifting the selectivity to isoparaffin. The conversion of n-heptane increased with an increasing temperature from 220 °C to 260 °C, which is possibly related to the dominant isomerization reaction at lower temperature[13].Simultaneously, it can be observed that the conversion of n-heptane over various catalysts reached a maximum value when the reaction temperature was between 260—280 °C. If the temperature exceeded beyond the range of 280—300 °C, the conversion of n-heptane would decrease with the rise of the reaction temperature. Contrary to the pyrolysis procedure, isomerization is an exothermic reaction, so low temperature favors the isomerization reaction. In this paper, 260 °C was chosen as the reaction temperature for n-heptane isomerization over the catalyst. Thus, another reason for the decrease in n-heptane conversion at higher temperature might be caused by the cracking reaction[20].
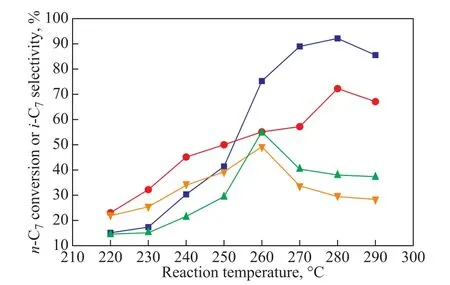
Figure 8 Influence of reaction temperature on the catalytic performance of Mg-MCM-48-0.02 and Zr-MCM-48-0.03(0.2 g of catalyst, reduction temperature = 400 °C, reduction time = 3 h, WHSV = 6.8 h-1)
3.2.2 Effect of WHSV
The effect of WHSV on isomerization of alkanes over M-MCM-48 catalysts is presented in Figure 9. Due to a similar trend, we chose Mg-MCM-48 as the object of analysis. It can be seen that when the weight hourly space velocity increased from 1.5 h-1to 13.5 h-1, the conversion of n-heptane decreased from 86.5% to 35.3%.This might be due to the following reasons: One reason is that the diffusion of the products in catalyst channels was difficult at lower space velocity, and the cracking rate was fast, which might lead to the low selectivity of isoheptane while the n-heptane conversion rate was higher; the other one was attributed to the shorter contact time between the reactant n-heptane and the catalyst at higher space velocity. In both of the two cases, the n-heptane conversion was decreased. The selectivity of the isomerization of the alkane could be improved by controlling the contact time of the reaction material with the catalyst to inhibit the generation of the multi-branched products and the occurrence of the cracking reaction,which could reduce the secondary reactions during the conversion of alkanes. The n-heptane conversion and the isoheptane selectivity were 36.03% and 75.71%,respectively, when the figure obtained at 6.8 h-1was chosen as the optimum weight hourly space velocity.
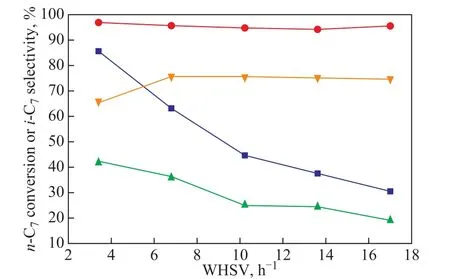
Figure 9 Influence of WHSV on the catalytic performance of Mg-MCM-48-0.02 and Zr-MCM-48-0.03 (0.2 g of catalyst, reduction temperature = 400 °C, reduction time = 3 h,reaction temperature = 260 °C)
It can be seen from the previous charts, Mg-MCM-48 showed a better catalytic effect no matter at the same reaction temperature or at the same reaction time as compared with the case of Zr-MCM-48. This research presented two aspects of reasons for this: On the one hand, it might be due to the fact that the amount of strong acid sites was higher for Mg-MCM-48 than other catalysts, so it could increase the activity of the catalyst;on the other hand, compared with Zr-MCM-48, Mg-MCM-48 had more lattice defects and heterogeneous surface. This fact could have a greater impact on the catalytic effect. The above two cases can be evidenced by the results of NH3-TPD, SEM and TEM analyses.
3.2.3 Effect of reaction time
Figure 10 shows the influence of the reaction time on the catalytic performance of Mg-MCM-48-0.02 and Zr-MCM-48-0.03 when the temperature was 260 °C. As regards Mg-MCM-48, when the reaction time was increased from 90 min to 130 min, the n-heptane conversion increased from 15.3% to 79.8%, while the selectivity to isoparaffin increased to a maximum value for 130 min and then decreased over a longer reaction time obviously. As for Zr-MCM-48, with the reaction time increasing from 90 min to 130 min, the n-heptane conversion increased from 21.3% to 49.8%, and the selectivity to isoparaffin increased to a maximum value for 130 minutes and then decreased over a longer reaction time.
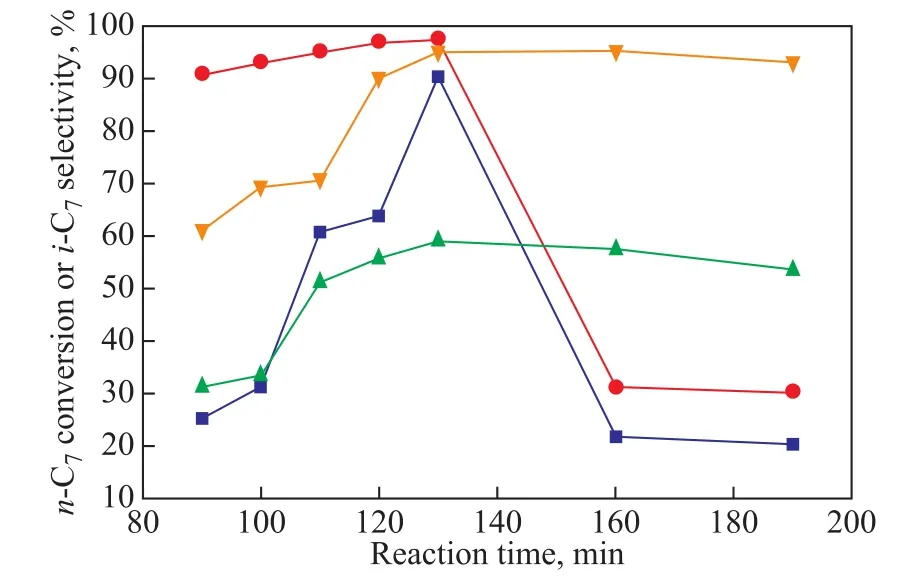
Figure 10 Influence of the reaction time on stream on the catalytic performance of Mg-MCM-48-0.02, Zr-MCM-48-0.03 (0.2 g of catalyst, reduction temperature =400 °C, reduction time = 3 h, reaction temperature = 260 °C,WHSV = 6.8 h-1)
When the reaction time was shorter than 130 min, more active metal components (Zr or Mg) could be reduced with the extension of the reaction time, therefore, there could be more active metal components (Zr or Mg) to cat-alyze the target reaction, and the conversion of n-heptane could be increased. The isomerization of n-heptane followed the classical bifunctional mechanism of alkane hydrocracking and hydroisomerization, which involved the(de)hydrogenation steps on the metallic sites and carbenium formation with rearrangements on the acidic sites[21-22].As for the Mg-MCM-48 or the Zr-MCM-48 bifunctional catalysts, the catalysis process could include the following steps: dehydrogenation of the n-heptane taking place on Mg or Zr sites; the carbenium formation with rearrangements on the acidic sites to form isoalkenes; and hydrogenation of isoalkenes to form isoalkanes which could also occur on Mg or Zr sites[23]. Besides, when the reaction time was shorter than 130 min, the reason for the relatively high isomerization selectivity of the catalyst could be attributed to the mild acidity, which was suitable for isomerization rather than cracking, and the structure of the three-dimensional helical channels of M-MCM-48 molecular sieve could restrict the formation of the macromolecular intermediates. The inhibition of the cracking reactions might occur, because the intermediate carbon positive ions, which were formed on the acidic sites,were attached to the metal ions timely[24]. As the reaction proceeded, the cracking products were deposited on the active sites of the catalyst, leading to a decrease in the conversion of n-heptane. However, the pyrolysis products were preferentially deposited in the acidic medium, and consequently the selectivity of the isoparaffins increased at the beginning of the reaction. So the reasons for the deactivation of the catalyst with the reaction time as shown in Figure 9 varied, among which the carbon deposition was one that could greatly reduce the activity of the catalyst[25]. In addition, the sintering of active metal phases was probably another reason leading to the reduction of catalyst activity[26-27]. Other reasons could also cause the deactivation of the catalyst, such as the blockage of the molecular sieve pores and the collapse of the long molecular sieve structure, as well as the loss of Br?nsted acid sites, etc.[28-29]It can be concluded from Figure 8 that the stability of Zr-MCM-48 was relatively higher than that of Mg-MCM-48.
3.3 Distribution of products
In the course of n-heptane isomerization, the analysis of products formed over the Mg-MCM-48 catalyst is demonstrated in Table 2. The products of n-heptane isomerization mainly consisted of 2-MH, 3-MH, 2,2-DMP,and 2,3-DMP with only a small amount of branched chain products (DMP), while the cleavage products were composed mostly of C3and C4alkanes. The ratio of the multi-branched heptane isomers and the mono-branched heptane isomers remained at about 0.8, indicating that the n-heptane hydroisomerization reaction over Mg-MCM-48 gave rise to the high-octane gasoline components. The ratio of the multi-branched to the mono-branched isoheptanes trended down with the increase of reaction temperature, denoting that the cracking of multi-branched isomers was easier than that of the mono-branched isomers.Because there are no hydrocarbons having the carbon number more than seven in the cracked products, it is inferred that the isomerization reaction is carried out mainly according to the mechanism of single molecular reaction,rather than the bimolecular reaction mechanism proposed by Blomsma[30].
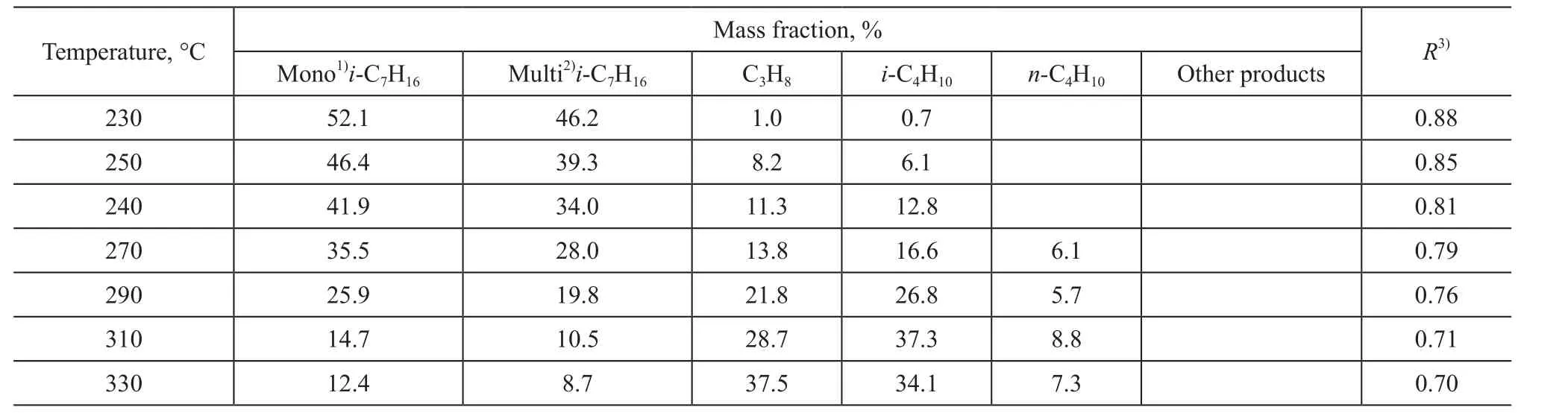
Table 2 Analysis of products formed over Mg-MCM-48 catalyst
This work has revealed that the mesopore-structured MCM-48 material used as the catalyst support is unusually appropriate to zeolite because of its greater shape selectivity in the skeletal isomerization of n-heptane. Two interpenetrating continuous networks of chiral channels,which are made up of large surface area, good long-range periodic and three-dimensional cubic Ia3D structure, can play a key role in the shape selectivity of hydrocarbons.Simultaneously, the strength of surface Br?nsted acidity of our catalysts is moderate, which is favorable to skeletal isomerization, because a too strong Br?nsted acidity can usually favor the cracking reaction, while a too weak acidity can result in low catalytic activity[31-32].
4 Conclusions
Highly ordered M-MCM-48 (where M = Zr, Mg) mesoporous materials were synthesized by the hydrothermal method. Compared with MCM-48, Zr (or Mg)-MCM-48 still maintained the typical cubic mesoporous framework of MCM-48, with a slight decrease of specific surface areas and mesopore orders. Both Zr-MCM-48 and Mg-MCM-48 exhibited weak and medium strong acid sites.Under comparable conditions, Mg-MCM-48 displayed a higher catalytic activity than Zr-MCM-48. The optimum reaction conditions of Mg-MCM-48 covered a reaction temperature of 280 °C and a WHSV of 3.3 h-1, along with a heptane conversion of 91.5% and a corresponding isomerization selectivity of 72.3%. However, Mg-MCM-48 exhibited a poorer stability than Zr-MCM-48.Mg-MCM-48-0.02 could maintain a relatively high catalytic activity for only 130 min at a reaction temperature of 260 °C and a WHSV of 6.8 h-1, while Zr-MCM-48-0.03 could maintain a relatively high catalytic activity for more than 190 min under the same reaction conditions.
Acknowledgement:This work was supported by the postdoctoral programme for chemical engineering and technology of Northeast Petroleum University, the Youth Fund of Northeast Petroleum University (grant number NEPUQN2015-1-08),the Cultivation Fund of Northeast Petroleum University [grant number 2017PYYL-03], the Daqing Science and Technology Plan Projects [grant number szdfy-2015-04], and the College Students’ Innovative Entrepreneurial Training Plan Projects of Heilongjiang Province [grant number 201710220030].
- 中國煉油與石油化工的其它文章
- Comparison of Lubricities of Two Novel Benzotriazole Derivatives Used as Additives in Water-Glycol Hydraulic Fluid
- Effect of Biodiesel Soot on Tribological Behavior of Liquid Paraffin
- Heat Transfer Investigation and Modeling of Heat Integrated Distillation Column
- Novel Control Structure Design of Differential Pressure Thermally Coupled Reactive Distillation for Methyl Acetate Hydrolysis
- Preparation of Sodium Cobalt Tetracarbonyl and Optimization of Process Conditions for Hydroesterification of Ethylene Oxide
- Bulk Ni-Mo Composites Prepared by Solid Reaction Method and Their Hydrodeoxygenation Performance

