Laboratory Safety of Dupilumab, and lts Effect on lnflammatory Biomarkers, in Chinese Adults With Moderate-to-Severe Atopic Dermatitis: An Analysis of a Randomized, Double-Blind Phase lll Study
Yan Zhao, Li-Ming Wu, Qian-Jin Lu, Xing-Hua Gao, Xiao-Hong Zhu, Xu Yao, Lin-Feng Li, Wei Li,Yang-Feng Ding, Zhi-Qiang Song0, Ling-Ling Liu, Ning-Ning Dang, Chun-Lei Zhang, Xiao-Ming Liu,Jun Gu, Jin-Yan Wang, Song-Mei Geng, Quan-Zhong Liu, Yi-Feng Guo, Li Dong0, Shelley Li0,Christine Xu0, John T. O’Malley0, Elizabeth Laws0, Nikhil Amin, Ashish Bansal, Min Wang,Jian-Zhong Zhang,*
1 Department of Dermatology, Peking University People’s Hospital, Beijing 100044, China; 2 Hangzhou First People’s Hospital, Hangzhou,Zhejiang 310006, China; 3 The Second Xiangya Hospital of Central South University, Changsha, Hunan 410011, China; 4 The First Hospital of China Medical University, Shenyang, Liaoning 110001, China; 5 Wuxi Second People’s Hospital, Wuxi, Jiangsu 214002, China; 6 Hospital for Skin Diseases, Institute of Dermatology, Chinese Academy of Medical Sciences, Nanjing, Jiangsu 210042, China; 7 Beijing Friendship Hospital, Capital Medical University, Beijing 100050, China; 8 Huashan Hospital, Fudan University, Shanghai 200040, China; 9 Shanghai Skin Disease Hospital, Shanghai 200040, China; 10 The Southwest Hospital of AMU, Chongqing 400038, China; 11 Peking University First Hospital, Beijing 100034, China; 12 Jinan Central Hospital, Jinan, Shandong 250013, China; 13 Peking University Third Hospital, Beijing 100191, China; 14 University of Hong Kong-Shenzhen Hospital, Shenzhen, Guangdong 518040, China; 15 Changhai Hospital of Shanghai,Shanghai 200433, China; 16 Ningbo No. 2 Hospital, Ningbo, Zhejiang 315099, China; 17 The Second Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Shaanxi 710004, China; 18 Tianjin Medical University General Hospital, Tianjin 300052, China; 19 Xinhua Hospital Affiliated to Shanghai Jiaotong University School of Medicine, Shanghai 200092, China; 20 Research & Development, Sanofi, Bridgewater, NJ 08807, USA; 21 Research & Development, Regeneron, Tarrytown, NY 10591, USA; 22 Medical, Sanofi China, Shanghai 200040, China.
Abstract Objective: Limited information is available on the use of dupilumab for the treatment of atopic dermatitis (AD) in the Chinese population.Methods: We analyzed laboratory data from a previously published randomized, double-blind phase III trial (NCT03912259)to provide further insight into the safety of dupilumab in Chinese adults with moderate to severe AD. The trial participants received either 300 mg of dupilumab or placebo every 2 weeks for 16 weeks. Hematology, blood chemistry, serum thymus and activation-regulated chemokine (TARC), and total immunoglobulin E (IgE) were evaluated.Results: In total, 82 participants received dupilumab and 83 received placebo. With the exception of eosinophil counts of>0.8 × 109/L, which were found less frequently with dupilumab (9.8%) than with placebo (18.7%), the hematology and blood chemistry values were generally stable in both treatment groups. There were no clinically significant differences between the dupilumab and placebo groups, and no participants developed treatment-emergent abnormalities of potential clinical significance. However, compared with placebo, greater decreases in serum lactate dehydrogenase (mean change, -97.4 vs.-33.5 IU/L), TARC (median percent change, -78.6% vs. -30.8%), and total IgE (median percent change, -53.4% vs. -0.2%)were observed with dupilumab than placebo at week 16.Conclusion: Dupilumab demonstrated a favorable laboratory safety profile in Chinese adults with moderate to severe AD.
Keywords: atopic dermatitis, dupilumab, laboratory safety, Chinese
lntroduction
Atopic dermatitis (AD) is a systemic inflammatory disorder that occurs as a result of epidermal barrier disruption and dysregulation of the type 2 immune response.1Clinically, AD is characterized by recurrent eczematous lesions and pruritus.1
Dupilumab is a fully human monoclonal immunoglobulin (Ig) G4antibody that inhibits interleukin (IL)-4 and IL-13 signaling. IL-4 and IL-13 are major drivers of the type 2 immune response and play key roles in the pathogenesis of AD, as well as in the pathogenesis of other atopic conditions such as asthma, allergic rhinitis, and food allergies, which are also common in individuals with AD.2-3
Dupilumab monotherapy was shown to be efficacious in reducing the signs and symptoms of AD over the course of 16 weeks in 2 randomized, double-blind, placebo-controlled, phase III studies: SOLO 1 and SOLO 2.4In addition, 1 year of treatment with dupilumab plus topical corticosteroids was efficacious in improving AD in the randomized, double-blind, placebo-controlled,phase III CHRONOS study.5In all 3 studies, dupilumab had an acceptable safety profile.4-5A pooled analysis of the SOLO 1, SOLO 2, and CHRONOS studies showed that treatment with dupilumab produced no clinically important changes in laboratory parameters, including hematology, serum chemistry, and urinalysis parameters.6Furthermore, no clinically meaningful changes in hematology, blood chemistry, or urinalysis parameters were noted in adults with moderate to severe AD who were treated with dupilumab for up to 3 years.7
However, only limited efficacy and safety data are available on the use of dupilumab in the Chinese population.To address this, a placebo-controlled clinical trial of dupilumab was conducted in Chinese adults with moderate to severe AD (NCT03912259). The main results, which were previously published,8showed that dupilumab was significantly more efficacious than placebo: 26.8%of the participants randomized to dupilumab achieved the primary efficacy endpoint (achievement of both an Investigator Global Assessment score of 0–1 and a reduction of ≥2 points between baseline and week 16), compared with only 4.8% of those randomized to placebo (P< 0.0001). Moreover, the incidence of treatment-emergent adverse events was similar in the 2 treatment arms.
The purpose of this article is to present the main laboratory safety findings of the trial as well as exploratory data on serum biomarkers of AD disease activity.
Materials and methods
Study design, recruitment criteria, and treatment
This was a randomized, double-blind, placebo-controlled,parallel-group, phase III trial (Clinicaltrials.gov identifier NCT03912259). The study design and methodological details have been previously described.8Briefly, Chinese adults with moderate to severe AD that was uncontrolled with topical medications, or for which such medications were inadvisable, were randomized to receive subcutaneous dupilumab (loading dose of 600 mg, followed by 300 mg) or matching placebo once every 2 weeks for 16 weeks. Follow-up was then performed for 12 weeks.
As detailed in the primary publication of this study,8concomitant treatment with the following was prohibited: live (attenuated) vaccines, immunomodulating biologics, topical calcineurin inhibitors, topical or systemic corticosteroids, systemic nonsteroidal immunomodulating or immunosuppressive drugs, or initiation or up-titration of antigen-specific immunotherapy. However,rescue treatment with otherwise prohibited agents was permitted at the discretion of the study investigators.
Outcomes
The primary efficacy and safety endpoints of the trial have been previously published.8The secondary endpoints of the study included laboratory (hematology and clinical chemistry) safety parameters, and these are the subject of the present report. Several biomarkers have been suggested for evaluating the level of AD severity, including lactate dehydrogenase (LDH), serum thymus and activation-regulated chemokine (TARC), and IgE.9-11The serum level of LDH has been shown to be positively correlated with the clinical severity of AD,9and LDH has thus been proposed as a potential serological marker of the therapeutic response.12-14LDH was therefore included as part of the laboratory safety plan in the present study. We also included the TARC and total serum IgE levels (at baseline and week 16) as exploratory endpoints to further our understanding of changes in AD-associated biomarkers during dupilumab treatment.
Hematology and clinical chemistry parameters were measured at baseline and at weeks 8, 16 (end of treatment), and 28 (end of study). For each laboratory safety parameter, we recorded the number and proportion of participants who had at least 1 treatment-emergent potentially clinically significant abnormality (PCSA). This was defined as an abnormal value that was considered medically important by the sponsor and that developed or worsened between the first dose of study treatment and the end of the on-treatment period.
Statistical analysis
Clinical laboratory parameters were assessed in the safety analysis population, which comprised all randomized participants who received at least 1 dose of the study treatment. Continuous variables are summarized as mean and standard deviation (SD) or median and interquartile range(IQR). Logistic regression was used to correlate the baseline TARC and IgE levels with the clinical efficacy endpoints. The odds ratio (OR) and 95% confidence interval(CI) were calculated for the likelihood of achieving the efficacy endpoints in participants with a high (above median)versus low (below median) level of IgE or TARC. The significance level was 0.05; however, calculatedPvalues were nominal. All statistical calculations were conducted using SAS?version 9.4 software (SAS Institute, Cary, NC, USA).
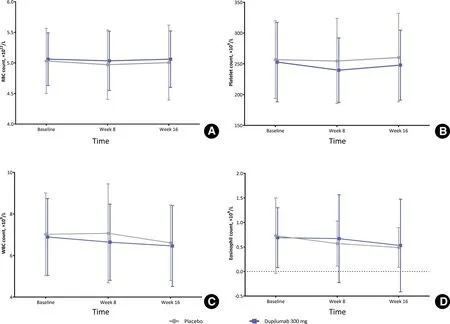
Figure 1. Hematology parameters (mean values) of patients with moderate-to-severe atopic dermatitis received 300 mg dupilumab and placebo over time. (A) RBC count. (B) Platelet count. (C) WBC count. (D) Eosinophil count. Error bars represent standard deviation. RBC, red blood cell; WBC, white blood cell.
Ethics
This study was conducted in accordance with the principles of the Declaration of Helsinki, the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use guidelines for Good Clinical Practice, and all applicable laws, rules,and regulations. All study participants provided written informed consent before any study procedures were conducted.
Results
In total, 165 participants were randomized; 82 participants received dupilumab and 83 received placebo.The participants’ baseline demographic and clinical characteristics have been previously published.8In brief, the 2 groups were well balanced at baseline; in the overall study population, the median age was 28.0 years, and 71.5% of the participants were male. Most of the participants completed the study treatment period (93% of dupilumab recipients and 80% of placebo recipients).8
The most commonly used concomitant medications by therapeutic class in either treatment group were systemic antihistamines (53.7% in the dupilumab group and 42.2% in the placebo group), antiallergic agents(34.1% and 31.3%), piperazine derivatives (26.8% and 20.5%), and antiallergic agents excluding corticosteroids (22.0% and 21.7%).
As previously reported,8rescue medication use was less common in the dupilumab than placebo group(any rescue medication [19.5%vs. 50.6% of patients],topical rescue medication [19.5%vs. 50.6%], and systemic rescue medication [2.4%vs. 4.8%]). Overall,tacrolimus was the most frequently used rescue medication (13.4% of dupilumab recipientsvs. 43.4% of placebo recipients), followed by topical corticosteroids(15.9%vs. 39.8%) and systemic corticosteroids (1.2%vs. 2.4%).
Hematology
The red blood cell, platelet, white blood cell, and eosinophil counts in the dupilumab and placebo groups atbaseline, week 8, and week 16 are shown in Figure 1.In addition, the hematology parameters at baseline and week 16 are shown in Table 1. At baseline, the mean (SD) platelet count was 252.8 (64.46) × 109/L in the dupilumab group and 256.8 (63.19) × 109/L in the placebo group. In both groups, the nadir of the platelet count was observed at week 8 (mean [SD]: 239.7[52.45] × 109/L for dupilumab and 254.6 [69.27] ×109/L for placebo). Compared with baseline, the mean eosinophil count decreased in both groups during the study. At baseline, the mean (SD) eosinophil count was 0.66 (0.610) × 109/L in the dupilumab group and 0.73 (0.771) × 109/L in the placebo group. At week 16, the median change in the eosinophil count from baseline was -0.20 × 109/L in the dupilumab group and -0.10 × 109/L in the placebo group. The proportion of participants who showed a shift from a normal to high eosinophil count was lower in the dupilumab group than in the placebo group at week 8 (7.4%vs.12.0%) and at week 16 (5.1%vs. 10.1%). The most common PCSA was an increase in the eosinophil count to >0.8 × 109/L (from ≤0.8 × 109/L at baseline). This occurred in 9.8% of participants in the dupilumab group and in 18.7% of participants in the placebo group. The incidence of all other hematology-related PCSAs was ≤5%.
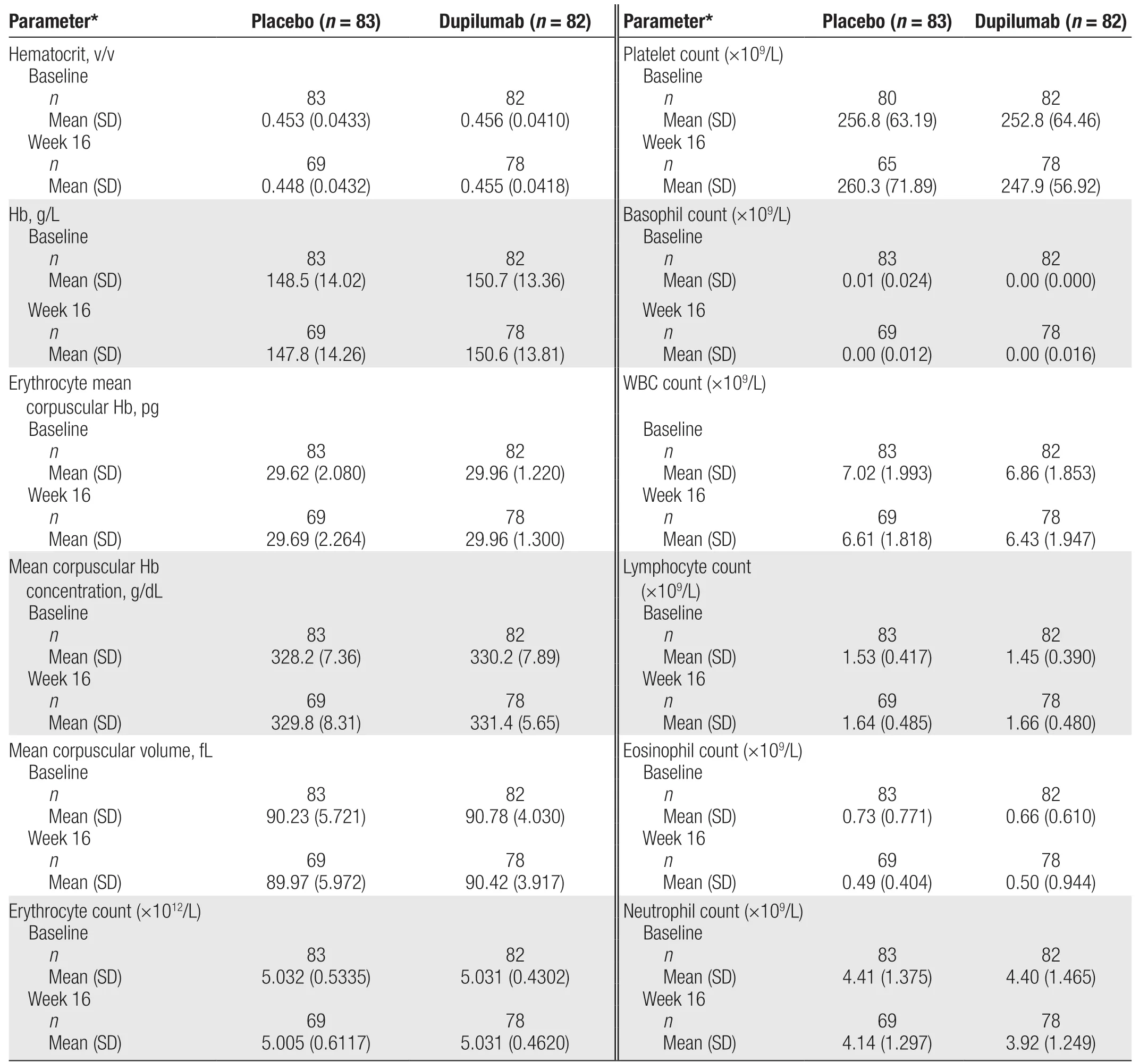
Table 1 Hematology parameters at baseline and week 16 in participants with atopic dermatitis who received dupilumab or placebo.
Serum chemistry

Table 2 Blood chemistry parameters at baseline and week 16 in participants with atopic dermatitis who received dupilumab or placebo.
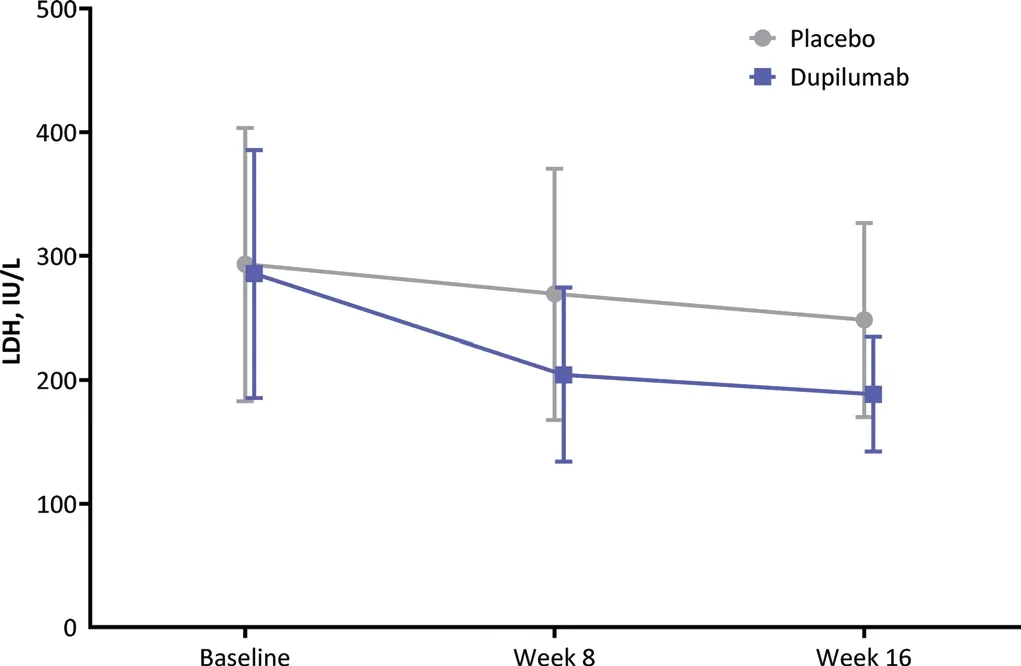
Figure 2. Mean LDH levels of patients with moderate-to-severe atopic dermatitis received 300 mg dupilumab and placebo over time.Error bars represent standard deviation. LDH: lactate dehydrogenase.
There were no meaningful differences in the mean change from baseline in any of the serum chemistry parameters(except LDH) between the 2 treatment groups (Table 2).Compared with baseline, the serum LDH concentration decreased at week 16 in both the dupilumab and placebo groups (Fig. 2). At baseline, the mean (SD)LDH concentration was 285.4 (100.03) IU/L in the dupilumab group and 293.2 (110.34) IU/L in the placebo group. At week 16, the mean (SD) change from baseline in the LDH concentration was -97.4 (77.3)IU/L in the dupilumab group and -33.5 (83.3) IU/L in the placebo group.
TARC and IgE
Participants who received dupilumab had a greater decrease in the TARC level compared with those who received placebo (Table 3). At baseline, the median (IQR)TARC level was 1,430.0 (621.0–4,940.0) pg/mL in the dupilumab group and 1,415.0 (622.0–5,270.0) pg/mL in the placebo group. At week 16, the median (IQR) percent change in the TARC level was -78.64% (-90.95%to -63.29%) in the dupilumab group and -30.79%(-56.47% to -5.33%) in the placebo group.
During the study, the total serum IgE level decreased in participants who received dupilumab and remained similar to baseline in those who received placebo (Table 3).At baseline, the median (IQR) total serum IgE level was 2,558.5 (438.0–8,611.0) IU/mL in the dupilumab group and 3,284.0 (527.0–10,381.0) IU/mL in the placebo group. At week 16, the median (IQR) percent change in the total serum IgE level was -53.36% (–63.83% to–43.84%) in the dupilumab group and -0.21% (–16.68%to 20.38%) in the placebo group.
When the probability of achieving the primary efficacy endpoint (defined in the Introduction) was analyzed according to whether the baseline TARC and IgE levels were high versus low, the OR for a high baseline TARC level was 0.3 (95% CI, 0.03–3.18;P= 0.3285)and that for a high baseline IgE level was 0.3 (95% CI,0.03–2.80;P= 0.2780). ThePvalues for interaction between the baseline TARC or IgE level (highvs. low) andstudy treatment (dupilumabvs. placebo), with regard to achieving the primary efficacy endpoint, were not statistically significant (P= 0.7220 for TARC andP= 0.7284 for IgE). The OR for achieving a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75) was 0.3 (95% CI, 0.07–1.13;P= 0.0730) for participants with a high versus low baseline TARC level and 0.2(95% CI, 0.06–0.98;P= 0.0465) for participants with a high versus low baseline IgE level. ThePvalues for the interaction between the baseline TARC or IgE level (highvs. low) and study treatment received (dupilumabvs. placebo), in terms of achieving EASI-75, were also not statistically significant (P= 0.1951 for TARC andP= 0.4427 for IgE). These findings indicate that patients with higher TARC and IgE levels at baseline were not more likely to respond to dupilumab, using either the Investigator Global Assessment or EASI as an efficacy measure, than those with lower levels. For details of the efficacy results of the trial, refer to Zhao et al.8

Table 3 TARC and IgE levels at baseline and week 16 in participants with atopic dermatitis who received dupilumab or placebo.
Other laboratory parameters
Blood chemistry and hematology parameters were similar in the dupilumab and placebo groups (Tables 1 and 2). There were also no clinically significant changes or between-group differences in urinary parameters (data not shown). The proportions of participants with treatment-emergent PCSAs were low and similar in the 2 groups (data not shown).
Discussion
The previously published results of this study showed that dupilumab was significantly better than placebo in improving the signs and symptoms of moderate to severe AD in Chinese adults.8In the present article, we showed that dupilumab had a favorable laboratory safety profile,relative to placebo, in this patient population. In general,the results of the blood chemistry and hematology assessments were stable during the study, and there were no clinically significant differences between the dupilumab and placebo groups. The incidence of PCSAs was also generally similar in the dupilumab and placebo groups with the exception of an eosinophil count of >0.8 × 109/L,which was more common in the placebo group. No clinically significant changes in other laboratory safety parameters were recorded in either group. Numerically greater reductions in the LDH, TARC, and IgE levels occurred with dupilumab than with placebo. Four participants in the dupilumab group had neutralizing antibodies, among whom 3 patients had low titers and 1 had a high titer.None of the participants in the placebo group had neutralizing antibodies.
The effect of dupilumab on laboratory safety parameters was previously investigated in a pooled analysis of the SOLO 1, SOLO 2, and CHRONOS trials, in which participants received dupilumab for up to 1 year,6and in an open-label extension study that included individuals who participated in phase I–III trials and in which dupilumab was administered for up to 3 years.7Both analyses showed that dupilumab produced no clinically significant changes in laboratory safety parameters in adults with moderate to severe AD.6-7The present study provides further evidence in support of the laboratory safety profile of dupilumab, showing similar results in Chinese participants with moderate to severe AD. Collectively, these data suggest that individuals treated with dupilumab do not require routine monitoring of laboratory safety parameters, which differentiates dupilumab from other systemic treatments for AD.15
Consistent with previous randomized controlled studies,6we found that treatment with dupilumab reduced the LDH level in Chinese patients. A reduction in the LDH level in individuals with AD has also been reported in real-world studies of dupilumab.16-17
An analysis of pooled data from SOLO 1, SOLO 2, and CHRONOS and a retrospective study of 61 Japanese individuals with moderate to severe AD both showed that the eosinophil count increased in some individuals treated with dupilumab.6,16In the current study, approximately 5% of participants in the dupilumab group and 10% in the placebo group showed an increase in the eosinophil count from normal to high at week 16, although the mean eosinophil count showed a small decrease in both groups. A long-term open-label extension study of dupilumab showed a trend toward an increased eosinophil count during the first 12 weeks of treatment but a subsequent decrease over the remaining 136 weeks of observation.7
In addition to the LDH level and eosinophil count, the TARC and IgE levels can be used to assess the level of AD activity. Dupilumab has been consistently shown to reduce the levels of these biomarkers, both in randomized controlled trials and in real-world studies.16-19Similarly,the present study showed greater decreases in the TARC and serum IgE levels in individuals who received dupilumab compared with those in the placebo group.A recent comprehensive review of evidence on potential biomarkers indicative of disease severity in AD conducted by the International Eczema Council graded the evidence for TARC as “very high” and that for IgE and LDH as“high.”20However, at present, it is difficult to select a single biomarker that is correlated with the severity of AD. Because of the diversity of disease phenotypes, it is likely that a set of biomarkers will be needed for both evaluation of disease severity (in combination with clinical assessment) and identification of which patients will benefit from targeted biologics.20
Our study has 2 main limitations. First, this randomized, placebo-controlled trial used stringent eligibility criteria, and as a result, there may be differences between the study group and the general population of Chinese adults with AD. Second, the participants were followed over a relatively short period. Conducting real-world studies with longer durations of follow-up will help to address these limitations.
In conclusion, no clinically significant changes or differences between dupilumab and placebo were observed in most clinical laboratory parameters. Dupilumab was associated with marked reductions in biomarkers of the type 2 inflammatory response, including TARC and IgE.Our findings suggest that routine monitoring of laboratory safety parameters is not required during dupilumab treatment for moderate to severe AD.
Source of funding
The phase III trial on which this subanalysis is based(NCT03912259) was sponsored by Sanofi. Medical writing assistance with the preparation of this article was also funded by Sanofi.
Acknowledgments
We would like to thank Georgii Filatov, Sarah Greig,and Richard Crampton of Springer Healthcare Communications, who provided medical writing support for this manuscript. This medical writing assistance was funded by Sanofi. Finally, we would like to thank the following individuals for contributing to patient enrollment: Zhi-Rong Yao, Dong Lyu, Hong Ren, Xiu-Min Yang, Dan-Qi Deng, Shan-Shan Li, Xiu-Ping Han, Zhen-Shu Biao, Min Zheng, Jian Hu, Yu-Xin Wang, Cong Yu,Guang-Dong Wen, Lin Cai, Ping Liu, Jiang Jin, Xue Chen,Xiao-Lan Ding, Zhang-Lei Cai, Jian Chen, Ming Su,Jian-Bo Zhong, Ya-Li Jia, Ying-Lian Cai, Meng-Hua Li,Jue Liu, Ping Yang, Ya-Fen Li, Ning-Yan Gu, Qing-Jie Hu,Jia Qi, Xun-Yi Dai, Xiao-Li Zhang, Ping-Dong Jiang, Hui Sun, Li-Jia Yang, Yan Hu, Mei-Shan Piao, Shi-Qin Tao,Ru-Sshan Xia, Jia Hu, Qi Wang, Wei Xu, Yan Li, Qin-Ping Yang, Shang-Shang Wang, Fang Yan, Hui-Bin Yin, Ning Yu, Zi-Wen Sun, Jia-Hui Zhao, Ruo-Jun Wang, Meng-Long Ran, Qian-Qian Li, Xue-Yan Lu, Yuan Cao, Qian Zhang, Zhen-Zhen Ye, Yu-Zhen Lan, Yi-Meng Wang,Jin-Zhu Guo, Guo-Jing Qin, Min Zhang, Zhi-Fang Zhai,Huan Wang, Chun-You Wang, Juan Wang, Su-Chun Hou,Li Ma, Xiao-Hong Chen, Tian-Meng Yan, Jia-Liang Shi,Jun-Hua Yan, Xin-Ling Bi, Yan Xue, Chang-Yuan Han,Ting Jiao, Li-Qin Liu, Xiao-Lu Wang, Xiang Xu, Guo-Bin Zhang, Yi-Ying Ye, Jin-Gang An, Long-Fei Zhu, Hui Zhang, Li Zhang, Li-Xin Xia, Rui-Qun Qi, Xiao-Ting Wen, Ping-Ping Qin, Long Hai, Shuai-Han-Tian Luo,Hui-Ping Wang, Quan Zhou, Xing-Ji Jin, Ying Wang, Min Sun, Ting-Ting Wang, Chun-Tao Xie, Wei-Wei Kuang,Qing Luo, Lin Li, Li-Mei Yuan, Yun Guo, Xiao-Lan Li,Yun-Feng Bi, Wen-Ting Cao, Xiao-Hong Zhou, Mei Cai,Hong Xie, Yuan-Yuan Liu, and Henan Si.
Data availability statement
Qualified researchers may request access to patient-level data and related study documents, including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient-level data will be anonymized and study documents will be redacted to protect the privacy of the trial participants. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at https://vivli.org.
- 國際皮膚性病學(xué)雜志的其它文章
- Research Progress of Genomic Variation in Psoriasis
- Perceptions of Acne and Its Treatments Among Chinese College Students: A Cross-Sectional Survey
- Sexual Behavior and Awareness of Sexually Transmitted Diseases Among Street-Based Female Sex Workers in the Florence Area, Central Italy
- Perspective on Melanoma in the Arab World:A Quantitative and Qualitative PubMed-Based Analysis of Research Output (2004–2019)
- Understanding the Pathogenesis of Generalized Pustular Psoriasis Based on Molecular Genetics and lmmunopathology
- Characterization of Generalized Pustular Psoriasis in Northwest China: A Single-Center Retrospective Study

