Subchondral bone microenvironment in osteoarthritis and pain
Yan Hu,Xiao Chen,Sicheng Wang,Yingying Jing and Jiacan Su
INTRODUCTION
Osteoarthritis(OA)is a series of diseases characterized by articular cartilage destruction and persistent pain.1The prevalence of OA is increasing,with an estimated number of patients of 250 million,and its risk factors include aging,sex,obesity,chronic systemic diseases,genetics,and injury.OA is believed to be a whole-joint disorder involving the bone,cartilage,synovium,ligament and joint capsule.The pathogenesis of OA involves at minimum mechanical factors,inflammation and metabolism,and these factors interact with each other,thus leading to poor curative effects for most treatments,except joint replacement at the end stage.
The initial pathological changes in OA have remained unclear until now,but it is certain that various pathologies associated with OA could worsen in combination,including cartilage degradation,subchondral bone sclerosis,angiogenesis and nerve innervation.Current therapies for symptomatic OA can be divided into several types:oral drug administration,intraarticular drug injection,intravenous drug administration and surgical operations.Generally,oral medications include NSAIDs and glucosamine;medicines injected into the joint space include sodium hyaluronate,glucocorticoids and anesthetics;and surgical options include but are not limited to total or partial joint replacement,arthroscopic surgery and high tibial osteotomy.Significantly,there is no single therapy that can completely resolve this disease with a complex origin,and further classification of OA subtypes based on their pathogenesis could be the only method of disease resolution in the future.As therapies aimed at articular cartilage or synovial components are not satisfactory and the mechanism of bonecartilage crosstalk is gradually becoming clear,we reviewed the critical role of subchondral bone in osteoarthritis and concluded that subchondral bone could be an ideal“entry point”for osteoarthritis treatment.
SUBCHONDRAL BONE MICROENVIRONMENT IN OA
Subchondral bone refers to the bony layer beneath the hyaline cartilage and cement line and can be divided into two parts anatomically,the subchondral bone plate(SBP)and subchondral bone trabecula.The SBP is a compact,polyporous calcified plate crisscrossed by multiple vessels and nerve fibers.Subchondral bone trabeculae are cancellous bony structures subjacent to the SBP that undergo continuous bone remodeling.Subchondral bone provides both mechanical and nutritional support for cartilage,indicating that microenvironmental changes in subchondral bone might affect cartilage metabolism,directly or indirectly.
Subchondral bone marrow lesions
A bone marrow lesion(BML),or so-called“bone marrow edema”2or“bone marrow edema-like abnormality”,3describes a noncystic,ill-defined,high-signal area on T2-weighted or proton densityweighted MRI.4BMLs are divided into the subchondral type and ligamentous type,5depending on the location.Subchondral BMLs(SBMLs),which result in abnormal signals on MRI beneath the calcified cartilage layer,affect more than half of asymptomatic individuals over 50,and their prevalence increases with age.6SBMLs can be observed in the early stage of knee OA and are thought to be helpful in early screening.
Interestingly,in approximately two-thirds of OA patients with SBMLs,cartilage erosion occurs around the superficial area of the SBMLs,5indicating the underlying linkage between SBMLs and OA progression.The worsening of SBMLs based on MRI manifestations is associated with subsequent radiographic findings and persistent pain.7–9At the site of an SBML,a high in situ turnover rate,pain sensitization and proinflammatory signaling activation have been observed through histological analysis and microarray techniques,10finally resulting in increased bone mineral density(BMD)and subchondral sclerosis.11Clinical observations have shown that over a period of 24 months,the size of the subchondral high-signal area on MRI increases,particularly in the medial tibia plateau and medial condyle,presenting a strong relationship with the loss of cartilage volume in corresponding regions.12Recent clinical data have shown the strong association between BMLs and cartilage damage in the tibial plateau,indicating the diagnostic value of predicting the degenerative status within the osteochondral unit.13
Subchondral bone cells around the chondro-osseous junction
Mesenchymal stromal cells(MSCs)are cell clusters with the capacity for self-renewal and the ability to differentiate into adipocytes,osteoblasts,chondrocytes,and myocytes in an appropriate microenvironment.During an in vivo experiment,transforming growth factor(TGF)-β3-induced MSC homing completely repaired the cartilage structure without xenogeneic cell implantation,14indicating that a robust subchondral bone microenvironment might support full-thickness articular cartilage regeneration.Evidence suggests that enriched MSCs derived from synovium,synovial fluid15or periarticular adipose tissue16also participate in cartilage regeneration in multiple pathological conditions,including OA.17Despite the participation of MSCs of multiple origins,the presence of spontaneous cartilage formation in full-thickness defects in vivo leads to better consequences,18suggesting that an intact subchondral bone environment might contribute to the self-repair process more than the MSC population.
As matrix resorption cells,osteoclasts act as“conductors”in subchondral bone metabolism and are involved in not only bone resorption but also type H vessel location and sensory nerve innervation.Tartrate-resistant acid phosphatase-positive(TRAP+)cells located around the chondro-osseous junction area exhibit lower cathepsin K(Ctsk)expression and fewer nuclei than those in the marrow space.19Erosion of the growth plate by endothelial cells during bone development mediates bone elongation,and the Ctsklowosteoclast subtype plays a critical role in inducing the side-to-side anastomosis of blind-ended vessels.These so-called“vessel-associated osteoclasts”show a high affinity for type H vessels,which is supported by the receptor activator of nuclear factor-κB ligand(RANKL)expression of endothelial cells and induces the anastomosis of type H vessels.
Bone-cartilage interaction
The tidemark,or hyaline-calcified cartilage interface,was first reported in 1984 and has been found to shift slowly toward the joint space during aging.20Material exchanges across the tidemark or cement line are critical for cartilage metabolism in the absence of capillaries inside hyaline cartilage.The chondrocyte extracellular matrix comprises nearly 70%–80% water,collagen and proteoglycans,and it is impermeable to anionic molecules owing to the negative potential of proteoglycans.Because of this characteristic and the increase in the proteoglycan concentration with depth,an anionic contrast agent creates a density gradient in the cartilage layer,21so contrast-enhanced CT could help to diagnose early OA since anionic media uptake is accelerated at sites of cartilage injury.22Cationic contrast agents also have potential in the diagnosis of cartilage damage on imaging based on the electrostatic attraction effect;using nonionic agents,the collagen concentration can be mapped on contrast-enhanced CT.23
Evidence has demonstrated the particular relationship of mutual regulation between cartilage and subchondral bone.Experiments on small molecular diffusion have revealed the existence of direct molecular signaling linking cartilage and subchondral bone,24and this crosstalk could increase in OA,suggesting that the subchondral bone microenvironment is involved in cartilage degeneration in OA.The inhibition of TFG-β in subchondral bone MSCs results in a protective effect on chondrocytes,indicating that TGF-β-induced osteogenesis plays an important role in bone-cartilage crosstalk.25Chondrocytes could participate in subchondral remodeling through the RANKL/RANK/OPG signaling pathway,and RANKL and OPG produced by chondrocytes could regulate bone metastasis in certain situations,but their specific roles remain unknown.26During the epiphyseal extension process,chondrocyte-derived OPG facilitates bone formation by blocking osteoclast maturation27–28through β-catenin signaling,29and the senescence-associated secretory phenotype(SASP),which is characterized by the secretion of chemokines and proinflammatory cytokines,accelerates OA progression.30Hypertrophic chondrocyte-derived vascular endothelial growth factor(VEGF)participates in the recruitment of MSCs and endotheliocytes,thus promoting bone formation,vessel innervation and cartilage resorption.31
Direct cell-to-cell contact has been observed in some pathological sections,32in addition to indirect molecular crosstalk.Interestingly,the tidemark is not a single line but a complex three-dimensional structure.CT has revealed that the cartilage layer penetrates the calcified layer and eventually reaches the subchondral bone plate in OA,33further indicating direct cell-tocell communication among chondrocytes,osteocytes,and osteoclasts.In an ex vivo coculture system,MSCs promoted matrix production and chondrocyte proliferation through a trophic-associated mechanism instead of chondrogenic differentiation.34-35Chemotactic factors released from MSCs recruit endogenous stem cells to cartilage damage sites and facilitate regeneration,36and exosomes derived from MSCs have demonstrated analogous effects in multiple in vivo experiments.37–40Moreover,subchondral bone osteoblasts derived from the zone of sclerosis in OA patients altered the chondrocyte phenotype in vitro and accelerated hypertrophic differentiation and calcification.41Taken together,these clues suggest that cartilage and subchondral bone regulate each other reciprocally through both cellular and molecular signaling.
Uncoupling of bone remodeling in the OA subchondral bone microenvironment
High turnover rate.Relatively balanced modeling and remodeling processes maintain the subchondral bone microenvironment.42Once the mechanical load changes or any other uncertain etiology occurs,the turnover rate of subchondral bone adjusts to adapt(Fig.1).An elevated osteoclast number has been observed in samples from both humans and mice with OA,accompanied by aberrant subchondral bony structures and mechanical support for the superficial layer.Cumulative evidence has shown that osteocytes and hypertrophic chondrocytes,rather than osteoblastic cells,serve as major suppliers of RANKL during the remodeling process.28,43Since osteocytes are a critical component in identification of the skeletal load,44abnormal mechanical force activates RANKL signaling and osteoclastogenesis,leading to aberrant bone remodeling and osteosclerosis.In addition,Dmp1-Cre;RANKLf/fmice presented a significant increase in osteoclasts in a tail-suspension experiment,demonstrating the role of chondroblastic RANKL signaling in osteoclastogenesis.Subsequent evidence has shown that RANKL expression and production are elevated in arthritis models.26,45Furthermore,soluble RANKL has a sufficiently small molecular weight to pass through subchondral bone plate cavities,28thus allowing it to play a role in osteoclast maturation.46
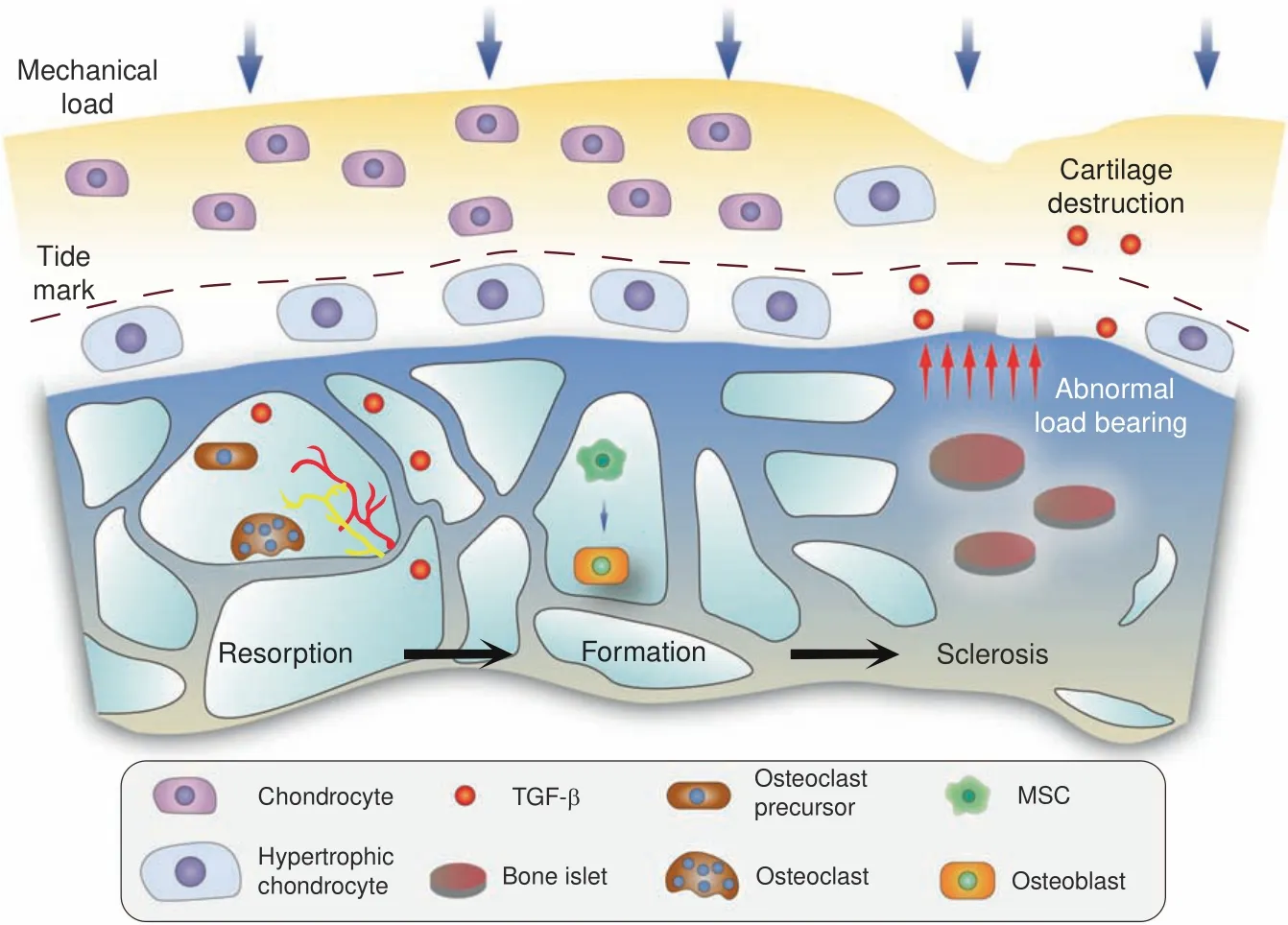
Fig.1 Schematic diagram of subchondral bone responding to aberrant mechanical loading and subsequent cartilage destruction.In the early stage of knee OA,bone resorption is overactivated,especially in the BML area.Biological factors,such as TGF-β,orchestrate MSC recruitment and bone formation,along with the generation of vessels and nerve fibers.Osteopetrosis and bone islets can be observed in advanced stages,which induce cartilage degeneration,at least in part,by abnormal load bearing
Bone marrow lesions were found in femoral head samples obtained from patients undergoing total hip arthroplasty,along with a high bone formation rate,hyperplastic woven bone and excessive angiogenesis.47Furthermore,a significant association between the subchondral bone turnover rate and cartilage damage has been proven by(99m)Tc-SPECT and 3-T MRI.48An elevated number and distribution of osteoclasts adjacent to the trabeculae are representative phenomena in SBMLs.49Subsequent remodeling processes are associated with angiogenesis and nerve growth,and the inhibition of osteoclast activity,angiogenesis or even nerve growth in subchondral bone marrow lesion sites might have potential therapeutic effects against both OA progression and pain.Siebelt et al.revealed that the subcutaneous injection of alendronate reduces both bone turnover and osteoarthritis progression.50The preemptive administration of alendronate before OA induction has been found to prevent subchondral bone turnover,cartilage degeneration and osteoarthritis pain better than early or delayed treatment after model establishment.51Taken together,these findings indicate that in OA,subchondral bone undergoes an aberrant remodeling process with an elevated turnover rate under the regulation of both mechanical and biological signals.
Microstructural changes.Beneath the hyaline and calcified cartilage,subchondral bone undergoes precise modeling processes to maintain a normal microstructure.52According to Wolff’s Law,the internal architecture of bone develops according to the magnitude and direction of the applied load.53Mounting evidence has indicated that microstructural changes occur during the course of OA,as determined by morphometric assessment methods54and shown in Fig.2.Compared with uninjured joints,more pronounced subchondral bone lesions are observed in joints with meniscus tears or ligament damage,55suggesting that microstructural changes might be considered a contributor to secondary disease caused by alignment issues.The subchondral bone plate changes prior to trabeculae in OA and restricts bonecartilage substance interchange via its internal properties,such as thickness and porosity.56Living osteocytes show persistent metabolic activity,including perilacunar/canalicular remodeling(PLR).57Osteocytes digest and reestablish the matrix around them through the secretion of matrix metalloproteinase(MMP),58cathepsin K(ctsK),59carbonic anhydrase 2(CA2)60and other enzymes.Interestingly,the elimination of MMP-13 in osteocytes while maintaining its expression in chondrocytes degrades the canalicular structure and accelerates degradation in OA.A comparison between a cadaveric control and subchondral bone with OA supports this conclusion,indicating an association between subchondral bone microstructure and OA progression.61Subchondral bone trabeculae include rod-like and plate-like structures according to their spatial morphology.62A quantitative CT analysis of human samples revealed a higher bone volume,thicker plate and lower trabecular rod/plate ratio on the medial plateau than on the lateral side.63In addition,excessive pressure in obesity-related osteoarthritis leads to lateral deformation of the articular cartilage and creates horizontal fissuring at the osteochondral interface,64based on the observation of human samples.Analogous subchondral bone microstructural changes occur in the patella during knee osteoarthritis,65as well as the talus in ankle osteoarthritis.Diabetes mellitus has been shown to be a risk factor for osteoarthritis,66and significant mechanical impairments are observed even in patients without OA pain or cartilage attenuation,67supporting the idea that subchondral bone microstructural damage might be an important predictor of OA progression.
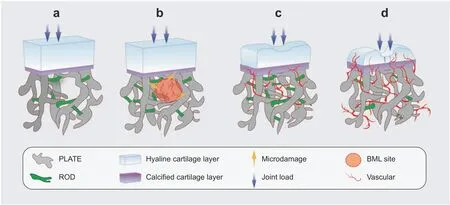
Fig.2 Subchondral bone microstructural transformation during OA progression.Rod-like and plate-like trabeculae are distributed accurately at a proper ratio to disperse stress.Osteogenesis coupled with angiogenesis lead to an increased BMD and conspicuous microstructural changes at the subchondral level.A decrease in the rod/plate ratio and subchondral bone plate thickening result in excessive mechanical support,which is detrimental to chondrocyte metabolism.Vessel-induced digestion of the cartilage matrix from the bottom up also takes part in the progression of cartilage destruction.a Normal state;b early stage with BML and vascularization;c progressive stage with cartilage damage;d end stage with severe cartilage destruction and vessel erosion
TGF-βsignaling.TGF-β1-3 is a functional growth factor secreted by multiple cells that are present only in mammals.The TGF-β superfamily participates in cell proliferation,differentiation,migration and apoptosis by sensing biological clues and mediating their signaling to serve as bioactive sentries.68TGF-β acts as an important growth factor regulating cell migration,the epithelialmesenchymal transition and other biological functions,and global defects in TGF-β lead to various major diseases,such as organ fibrosis and musculoskeletal disorders,including cartilage destruction.After secretion,TGF-β is normally stored in the extracellular matrix in a latent form,69or with so-called latency-associated peptide(LAP),and it can be cleaved into the mature form and latency-associated peptide in the proper environment.This inactive form enables the spatial and temporary activation of TGF-β for diverse physiological needs.Several TGF-β activators have been identified to date,most of which are markers of extracellular matrix(ECM)disorders,such as MMP,70plasmin,71and thrombospondin-1(TSP1),72as well as an appropriate pH.73TGF-β1 is abundant in the bone matrix,cartilage and skin,where it regulates the physical activities of these organs or tissues.74During bone resorption,a mildly acidic environment is generated around the resorption site at a pH of 4.5,releasing mature TGF-β1 and thus launching remodeling and modeling processes.75
Activated TGF-β binds specific receptors,named receptor I and receptor II;subsequent signaling is dependent on their complex forms and occurs mainly via Smad-mediated or Smadindependent pathways.68Stockpiles released from the bone matrix orchestrate MSC migration and differentiation and repair of the resorptive space.Specifically,TGF-β1 recruits MSCs in a gradient-dependent manner,76while other factors,such as IGF-1,77induce osteoblastogenesis.TGF-β also facilitates tissue repair by increasing MSC proliferation,mobilization and migration.The tissue regeneration process requires proper migration and accurate differentiation,and previous studies have reported that colonic crypt,78hair follicle79and renal fibrosis80repair could be accelerated by TGF-β.In a rabbit humeral cartilage defect model,cell-free scaffolds carrying TGF-β3 recruited significantly more MSCs than the negative control and achieved full regeneration without exogenous stem cell implantation.14In addition,TGFβ-regulating signaling also plays a role in the PLR by maintaining homeostasis of the osteocyte matrix.81
Aberrant TGF-β activation and subsequent signal transduction lead to multiple disease phenotypes,such as osteoarthritis(Fig.3),enthesopathy,82and Camurati-Engelmann disease(CED).The mutation of TGF-β1 causes CED,which is characterized by thickened cortical bone and narrowing of the marrow space83due to excessive TGF-β1 activation.A high incidence of osteoarthritis was observed among CED patients in former clinical research,84along with elevated active TGF-β levels.Additionally,the inhibition of subchondral TGF-β delayed OA progression,and knockout of the TGF-β type II receptor on MSCs achieved a similar effect in vivo.25Overall,subchondral osteocytes sense aberrant mechanical stress,induce osteoclast maturation through RANKL/RANK signaling,43and release TGF-β and other bioactive factors via both osteoclast resorption and the PLR process.Subsequently,endogenic MSC migration and unexpected bone islet formation disrupt mechanical homeostasis below the cartilage,which promotes chondrocyte apoptosis and substrate degradation.Anomalous hyaline and calcified cartilage place exceptional pressure on the subchondral region,further amplifying the force-induced turnover rate and creating a positive feedback loop to accelerate OA progression.Interestingly,mechanical force application could break the latent formation of LAP and release activated TGF-β,85which indicates that abnormal mechanical stress might play a role in cartilage TGF-β signaling activation,thus disturbing chondrocyte metabolism.Hypervascularization.Osteogenesis and angiogenesis are precisely coupled via type H vessels and multiple cytokines in bone metabolism.86Recent evidence has revealed a positive feedback loop in MSCs and type H vessels.87Platelet-derived growth factor receptor(PDGFR)-positive mesenchymal cells are spatially consistent with type H vessels,and angiogenic endothelial cells recruit MSCs through chemotaxis and promote osteogenesis.88Key signaling pathways in MSCs coupled with endothelial cells include the TGF-β,89PDGF-PDGFR,90angiopoietin,91Notch,92and FAK signaling pathways.93Thus,blocking the interaction between aberrant osteogenesis and angiogenesis might have a therapeutic effect on excessive subchondral osteosclerosis(Fig.4).
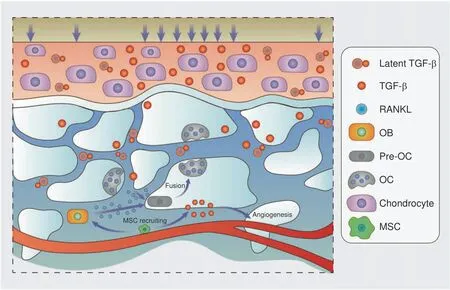
Fig.3 TGF-β signaling in the osteochondral area in OA.Shear force breaks the spatial structure of latent TGF-β,thus releasing activated TGF-β and accelerating bone remodeling.On the other hand,overactivated osteoclasts enable TGF-β expression,recruit MSCs and facilitate angiogenesis.In turn,bone and vessel formation promote cartilage TGF-β mobilization,and the cycle continues
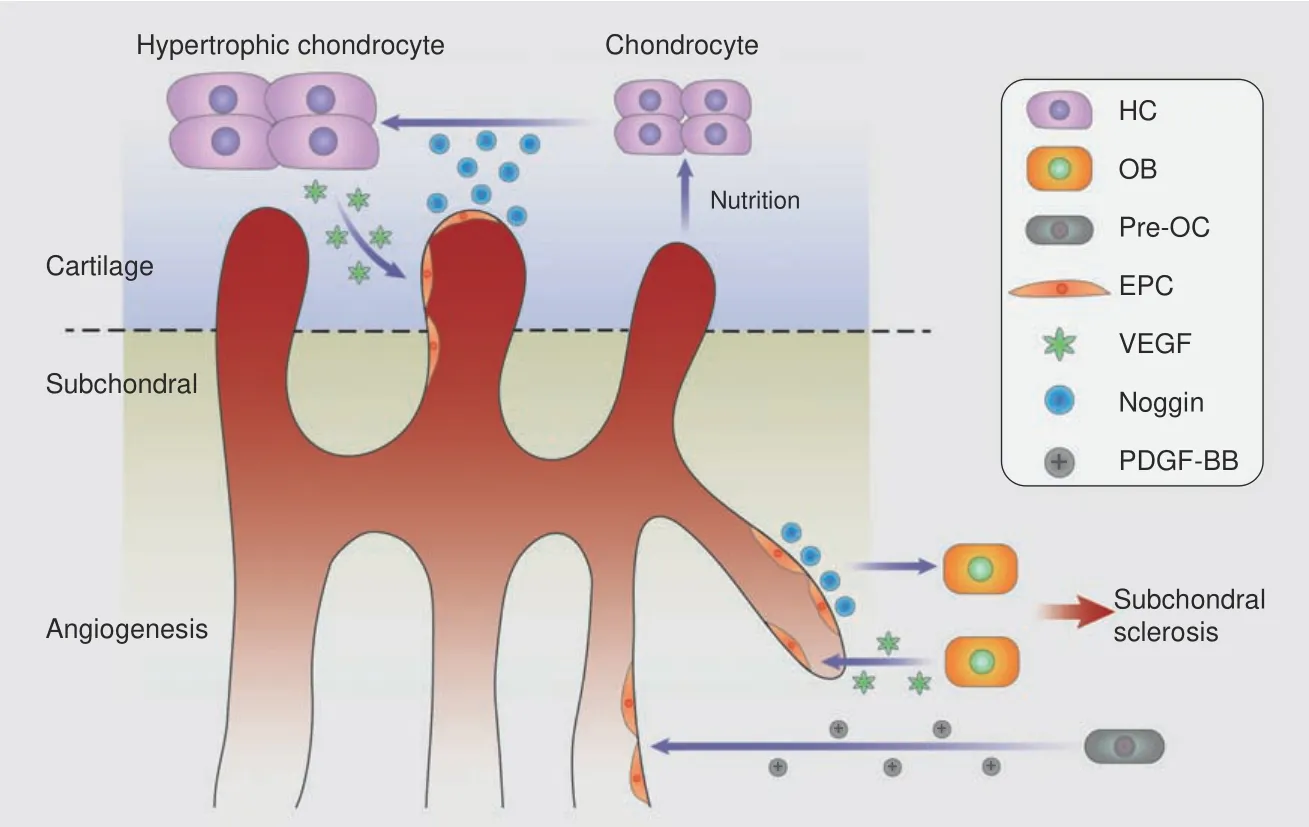
Fig.4 Coupled angiogenesis and osteogenesis in the subchondral bone accelerate cartilage destruction.Activated TGF-β and PDGF-BB derived from pre-OCs lead to neovascularization,type H vessels couple angiogenesis and osteogenesis by Notch signaling,and OBs accelerate this cycle via RANKL and VEGF release.Endothelial cells digest cartilage and contribute to chondrocyte hypertrophy,which in turn attracts vascular invasion by VEGF signaling
Hyaline cartilage is a nonvascularized tissue with resistance to vessel formation,but angiogenesis plays a critical role in endochondral bone formation,94–95suggesting that vessels might be responsible for cartilage digestion.Elegant research presented by Ramasamy et al.19proved the proteolytic function of endothelial cells,which misdirected the orientation of growth plate type H vessel elongation in the proximal tibia.Hypertrophic chondrocytes are a major source of VEGF,which links cartilage resorption,ossification and angiogenesis.96VEGF derived from osteoblasts can recruit osteoclasts and stimulate osteogenesis and angiogenesis,31and the effects of VEGF in the joint space can be blocked with an antagonist to hinder repair by repelling vascular invasion.97–98For maintenance of the avascular environment,chondromodulin(Chm-1),99TSP-1100and troponin 1(Tn1)101have been shown to counter cartilage angiogenic factors,such as VEGF,fibroblast growth factor(FGF)and TGF-β.
Resistance to vascular invasion in the joint decays in OA,and the spatial orientation of protease inhibitors makes them unable to expel vessels around the tidemark.Duplication of the tidemark and the penetration of vessels and nerve fibers have been observed in clinical OA samples via both micro-CT and highfrequency ultrasound.102Both angiogenic and antiangiogenic factors are overexpressed in OA chondrocytes,but protease inhibitors remain at normal levels in the deep layer of cartilage adjacent to the bony plate,103which might allow bottom-up neovascular invasion.In fact,the number of cartilage-resorptive type H vessels has been shown to be increased in subchondral bone in mouse models and human patients.Activated OA chondrocyte-derived mammalian target of rapamycin complex 1(mTOR1)signaling stimulates vessel invasion partially through the upregulation of VEGF-A expression,while nutrition optimization in capillaries in turn promotes mTORC1 activation.104Sensory nerve innervation.Nerve growth is normally coupled with angiogenesis under both physiological and pathological circumstances,105and perivascular cells stimulate nerve growth and help new axons navigate to their destination.Nerve fibers appear nearly two weeks after angiogenesis in rodent models.106Nerve growth factor(NGF)has been proven to stimulate both vascular and nerve growth in subchondral bone in OA and thus link angiogenesis and pain in the joint in OA.107Specifically,chondrocyte-derived NGF expression increases in the OA microenvironment and upregulates the expression of fibroblast growth factor in a dose-and time-dependent manner,thus promoting the angiogenic potential of endothelial cells.108Nerve growth and maintenance are also related to pleiotropic factors,such as VEGF and TGF-β,which are highly expressed in osteochondral junction areas in OA.Four types of axon guidance molecules,including netrins,slits,ephrins and semaphorins,are responsible for directing fiber extension and distribution.Both axon guidance molecules have unexpected angiogenic effects,109while perivascular cells,such as osteoclasts,regulate OA pain manifestation through axon guidance factor expression.
Sensory nerves sprout across bone fracture sites during normal healing,110which indicates a self-protection mechanism to prevent untimely weight bearing by injured limbs.Abnormal innervation occurs during all stages of the pathology of OA and is the main cause of continuously aggravated osteoarthritis pain.At an early stage,BMLs can be detected on MRI and occur along with the upregulation of neurogenesis-associated genes,10such asG protein-coupled receptor 158(Gpr 158),which has a neurotransmitter binding function,and ATPase H+-transporting lysosomal protein(ATP6V0D2),which plays a role in not only neuron projection but also osteoclast-osteoblast interactions.111–112Neovascular invasion along nerve fibers breaching the tidemark has been observed in clinical samples,while similar pathological changes are present in osteophytes in knee OA.113Calcitonin gene-related peptide-immunopositive(CGRP-IP)sensory nerve innervation has been shown to be significantly associated with OA pain manifestation,with the percentage of subchondral bone plate cavities containing CGRP-IP sensory nerves being obviously higher in the human symptomatic OA group than in the asymptomatic group.114
Taken together,these findings indicate that the microenvironment of subchondral bone in OA could be characterized by an elevated bone turnover rate,abnormal microstructures,overactive TGF-β,hypervascularization and aberrant sensory nerve innervation.These pathologies interact with each other and facilitate OA progression separately or in concert.
SUBCHONDRAL BONE MICROENVIRONMENT AND CARTILAGE DEGENERATION
Coupling of angiogenesis and osteogenesis contributes to cartilage erosion
Accumulating evidence suggests that bottom-up vascularization from subchondral bone plays a larger role than top-down vessel invasion originating from synovial tissue or synovium during cartilage erosion in OA.Matrix-digesting proteases,such as MMPs,act as“road sweepers”for the neovasculature,and inhibitors purified from cartilage called tissue inhibitors of metalloproteinases(TIMPs)are believed to help maintain the avascular status.115In the superficial cartilage layer,TIMP-1-positive chondrocytes have been found to be more abundant in OA samples than in the negative control,but in the deep zone near the bony plate,there is no significant difference.103This means that vascular resistance in the superficial layer of cartilage remains better than that in the deep layer in OA,suggesting a pivotal role of subchondral angiogenesis during cartilage destruction.
Endothelial cells erode the cartilage matrix during bone elongation,thus creating space for substantial osteogenesis.19,95Specifically,endothelial cells present 40-fold increased MMP-9 expression compared with osteoclasts during entochondrostosis,and the conditional knockout of MMP-9 in the postnatal endothelium leads to osteogenic defects and abnormally large growth plates,suggesting a critical role of endothelial-derived MMP-9 in cartilage resorption.In addition,the intra-articular administration of VEGF can accelerate OA progression in rodent models,116while anti-VEGF treatment can ameliorate cartilage degradation.117These data further indicate the cartilage resorption effect of neovascularization in both osteoarthritis and endochondral ossification.
Important research performed by Xie et al.has shown the critical role of preosteoclast-derived PDGF-BB in type H vessel genesis and bone remodeling.118The bone turnover rate is accelerated in the subchondral bone in OA,especially in BML zones;47the overactivation of osteoclastogenesis generates high levels of angiogenic factors,such as PDGF-BB and VEGF,and in turn,CD31+Endomucin+type H vessels accelerate the turnover rate and aggravate OA progression.119Inflammatory factors in the joint space,such as TNF-α,alter subchondral bone angiogenesis and MSC migration by inducing the expression of a newly discovered pathological angiogenesis regulator,leucine-richalpha-2-glycoprotein 1(LRG1).120Consistent with the higher clinical incidence of OA in females,recent literature has reported a more pronounced change in serum markers of neovascularization in female OA patients than in male OA patients.121
Abnormal mechanical support and cartilage metabolism
Mechanical stimulation and cartilage homeostasis.Articular cartilage needs proper mechanical loading to remain healthy,122and both overloading and underloading can be detrimental.Underloading,or even nonweight-bearing,leads to cartilage atrophy or degradation,123–124while overloading,which is most often caused by obesity or deformity,results in cartilage fissures,64bone sclerosis and osteoarthritis.125–126A recent multicenter data analysis has shown that static loading,mainly from body weight,and cumulative loading,such as that from excessive daily walking,are associated with the worsening of cartilage damage,especially in the medial tibiofemoral joint.127In the acute stage of cartilage impact experiments,mitochondrial dysfunction presents as an early response,which is followed by chondrocyte death,but chondrocytes from weight-bearing areas are more resistant to impact-induced cell death.128According to Song et al.,the gremlin-1-NF-κB signaling pathway plays an essential role in excessive load-induced cartilage degeneration.129Proper biomechanical stimulation not only facilitates matrix synthesis by chondrocytes,including matrix proteins,collagen and glycosaminoglycan,130but also helps maintain hydraulic permeability for necessary substance exchange.131–132Instead of physiological stimulation,excessive mechanical loading upregulates p38 phosphorylation and MMP-13 expression,133thus inducing matrix degradation and proteoglycan loss.134Moreover,subchondral bone perforations containing neovasculature and nerve fibers are colocalized with mechanical stress points,135indicating that biomechanical signals influence not only cartilage metabolism but also subchondral bone plate remodeling.MSC biological behavior under biomechanical signals.In vitro,MSCs can respond to mechanical stimulation and differentiate toward various lineages without altering the culture medium.136Ex vivo experiments have shown that chemoattractant-induced MSC migration is restrained under the influence of extrinsic force,which inhibits autonomous tissue repair.Subsequently,in the advanced stage of injury,cell density loss and matrix erosion can be observed by histology.128Under elevated cyclic compression in vivo,osteogenic differentiation increases as a result of various actions.MSCs under abnormal stress demonstrate an increased angiogenic capacity,and conditioned medium from mechanically stimulated MSCs results in the significant promotion of angiogenesis compared with that from the negative control.137Multiple cytokines detected in the abovementioned medium are closely related to vascular formation,such as FGF,TGF-β and MMP-2.Other research has indicated that osteogenic differentiation is promoted by BMPdependent signaling because it can be reversed by rhNoggin administration.138Therefore,MSCs in sclerotic subchondral bone could exhibit an excessive angiogenic ability,which facilitates cartilage degeneration,as mentioned above.
OA subchondral microenvironment and chondrocyte senescence Senescent phenotypical features are normally observed in chondrocytes in OA,such as upregulated β-galactosidase(β-gal)activity,upregulated cell cycle inhibitor p16INK4aexpression,shortened telomeres and SASPs.139Interestingly,the transplantation of aging chondrocytes into a healthy knee joint leads to the OA phenotype,including severe cartilage destruction,140which indicates an underlying role of chondrocyte senescence in the onset and progression of OA.In addition,epigenetic alterations have been revealed in aged human OA samples.141Chondrocyte senescence is closely related to the diseased microenvironment in subchondral bone.p16INK4apositive senescent MSCs have been identified in subchondral bone trabeculae in an anterior cruciate ligament transection(ACLT)-induced OA model,as well as in an aging mouse model(19–20 months old)142and a spontaneous OA mouse model.143Senescence-associated phenotypical features of MSCs are observed in subchondral bone in OA,including G0/G1-phase stagnation,increased β-gal expression,and decreased S-phase entry.144
Given the presence of senescent chondrocytes and subchondral pathologies in OA,we further investigated the intrinsic association between the subchondral microenvironment and cartilage senescence.SASPs derived from senescent or hypertrophic chondrocytes recruit immature osteoclasts and facilitate maturation.Moreover,exosomal microRNA from osteoclasts promotes hypertrophic transformation via Smad2 inhibition.145In addition,senescent MSCs derived from synovium and subchondral bone in OA have been proven to be detrimental to cartilage homeostasis,both in vivo and ex vivo.Overall,senescence can be identified as a global pathological characteristic that affects all participants in the OA microenvironment.SASPs from multiple sources affect proliferation,differentiation and other cell functions.However,further studies are required to elucidate the underlying mechanism.
SUBCHONDRAL BONE MICROENVIRONMENT AND OA PAIN
Mechanism of OA pain
It is a common complaint of orthopedists that osteoarthritis pain is not associated with the radiographic presentation(Fig.5).Recent work has shown a relatively weak relationship between cartilage loss and OA joint pain.During a 36-month clinical survey,a loss of cartilage thickness of 0.01 mm over 2 years was responsible for an increase of 0.32 in the WOMAC score,146indicating that the deep pathogenesis of OA pain is unclear and that further consideration might be necessary when dealing with worsening OA pain.Rather than cartilage,the abovementioned CGRP-IP nerve fibers in the osteochondral plate channels have been found to be more critical to OA pain in both humans and rats.114
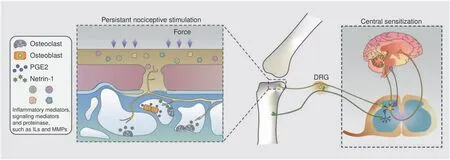
Fig.5 Schematic diagram of OA pain.Osteoclast-derived netrin-1 guides sensory nerve innervation in the osteochondral area,and multiple signaling mediators give rise to persistent nociceptive stimulation.The continuous input of pain signals causes central sensitization,suppresses the descending inhibitory system,and brings sympathetic referred pain away from the area of the arthritis lesion
Pain is often divided into three major types:nociceptive pain,which is caused by tissue damage in situ;neuropathic pain,which is caused by sensory nerve lesions;and idiopathic pain,whose etiology cannot be clearly explained.147The classification of pain in OA patients has long been neglected even though NSAID administration does not work for all patients complaining of stubborn joint discomfort.148In fact,neuropathic pain is more likely to react to nonstandard analgesics,such as tricyclic antidepressants.Recently,there has been growing evidence of the neuropathic component of OA pain,149and a deeper understanding of the pain mechanism could help in dealing with persistent joint pain.In contrast to structural changes in bone,cartilage and synovium,pain is a subjective phenomenon;thus,psychological factors may be involved,which cannot be ignored.
Nociceptive pain in situ
Around the knee joint,there are sensory and sympathetic nerve fibers that control nociception and proprioception.150As articular cartilage functions without internal vessels and nerves,nociceptive pain is naturally related to peripheral tissues,including the subchondral bone,meniscus and synovium.Experimental OA pain can be eliminated by the selective targeting of nociceptive primary afferents,151confirming the existence and therapeutic potential of nociceptive pain in osteoarthritis.Joint space inflammation plays a key role in OA chronic pain and acts as an initiator of cascade reactions.For example,synovial serine proteinase activates a group of G protein-coupled receptors called PARs and leads to several chronic pain conditions.152Neuropeptide substance P(SP),a neurotransmitter in the mammalian central and peripheral nervous systems,153has been found to be an injury-induced factor154that plays a critical role in the pathogenesis of OA.As part of the neurokinin-1(NK1)pain pathway,SP levels are elevated in both cerebrospinal155and synovial fluid156in OA patients with pain.The inhibition of SP-NK1 signaling with NK1 receptor antagonists results in partial anesis of nociceptive pain.157Growth factors and cytokines also participate in OA pain via both cartilage degradation and nociceptive stimulation.Pain relief has been observed for therapies targeting inflammatory mediators such as TNF,158IL-1 β,159IL-6160and PGE2,161signaling mediators such as p38,162HIF163and RUNX2,164and proteinases such as MMP165and ADAMTS-5.166
Neuropathic pain and sensitization in subchondral bone in OA
A systematic review has shown a 23% incidence of neuropathic pain among knee or hip OA patients,with considerable heterogeneity,167and gabapentin has been suggested to be helpful in dealing with OA pain.168Nerve fiber lesions can be tested by the presence of positive and negative signs.169Positive signs include hyperalgesia and allodynia,while negative signs include dysesthesia,numbness and a loss of vibration perception.The peripheral sensory nervous system transforms into a hypersensitive state,which can be activated by unproductive stimuli,and this supersensitive condition begins in the first few hours after injury or inflammation.170Both in vivo electrophysiological171examinations and subjective questionnaires for quantitative sensory testing147have revealed peripheral sensitization in OA patients or animal models.While most peripheral nociceptors remain silent,inflammatory factors lower their thresholds in the OA environment,172and nociceptive signals are transmitted through ascending pathways.
Constant nociceptive signals are transmitted by afferent neurons and are received by interneurons through synapses in the dorsal horn of the spinal cord,resulting in prolonged hyperexcitability150or so-called central sensitization in OA.Quantitative sensory testing(QST)of the pressure pain threshold(PPT)has confirmed the existence of central sensitization among OA patients.A low PPT value in the OA area indicates peripheral sensitization,while central sensitization presents as abnormal PPT values in remote areas.173Central sensitization directly leads toseparation of the nociceptive activation site and the pain location,which happens to be an osteoarthritis characteristic.174The precise mechanism of neurosensitization remains unclear,but successful total hip175or knee176arthroplasty can reverse this sensory abnormality,which indicates that periarticular injury or stimulation might be responsible for central nervous system hypersensitivity.In addition,descending inhibitory pathways in OA patients are weakened by multiple elements.Descending neural signals are disrupted in OA animal models,and medications such as norepinephrine reuptake inhibitors or opioid analgesics might help relieve intense OA pain.Tapentadol,an analgesic molecule that acts via both mechanisms,has been proven to be helpful against neuropathic pain in knee OA.177
Abnormal sensory nerve innervation links nociceptive and hypersensitive pain in OA
Elegant work has revealed aberrant nerve innervation and regulation in OA.178One week after ACLT surgery,invasion of the nociceptive sensory nerve near the chondro-osseous region could be observed by CGRP immunostaining,while the number of TRAP+cells and CGRP concentration were unaltered in the control group.This alteration,which was reflected by the number and density of nerve endings,was worse 8 weeks after surgery.
As previously described,the bone turnover rate and osteoclastogenesis are upregulated in subchondral bone,and the axon guidance molecule netrin-1,which is secreted by subchondral TRAP+cells,promotes and guides sensory nerve axon growth through the DCC receptor.Analogous sensory innervation induced by osteoclasts and netrin-1 has been observed in porous endplates and mediates spinal hypersensitivity.161This evidence explains the occurrence of pain relief after alendronate administration in certain areas in OA patients.
Overactive bone remodeling in subchondral bone is associated with bone sclerosis,angiogenesis,microstructural disruption and sensory innervation.In addition to the osteoclast-derived netrin-1-induced axon distribution,prostaglandin E2(PGE2)secreted by osteoblasts upregulates Nav1.8,a voltage-gated sodium channel expressed in dorsal root ganglion neurons and fibers,thus inducing hypersensitivity in the peripheral nerve environment.179Specifically,PGE2 and its receptor EP4 stimulate the phosphorylation of protein kinase A(PKA)and cAMP element-binding protein(CREB1),thus enhancing Nav1.8 transcription in dorsal root ganglion(DRG)neurons.Semaphorin 3A(Sema3A),derived from neurons,regulates bone remodeling in an osteoblast-independent manner,180and peripheral sensory nerves stimulate bone formation through Wnt signaling.181This evidence indicates the feedback regulation of sensory nerves in bone remodeling.In fact,PGE2 expression is significantly higher in areas of decreased bone density,such as in an osteoporotic mouse model.The peripheral sensory nerve detects the bone density by sensing the PGE2 concentration and stimulates the phosphorylation of CREB1 in the hypothalamus,thus maintaining the bone mass by regulating sympathetic nerve activity.182This evidence reveals the elaborate feedback loop involving subchondral bone remodeling,nerve innervation and hypersensitivity,which could be potential targets of future interventions for OA.
TARGETING OF THE SUBCHONDRAL BONE MICROENVIRONMENT FOR OA AND PAIN
Given the heterogeneity of symptoms and reactions to medications among OA patients,personalized treatment should be administered after complete assessment of the pathology according to the guidelines for different situations.Regarding the timing and method of surgery for end-stage OA,we need to be aware that total or partial arthroplasty might not allow complete remission to be achieved in all patients.183In addition to various surgeries,an etiological analysis and targeted therapies might improve the prognosis(Table 1).
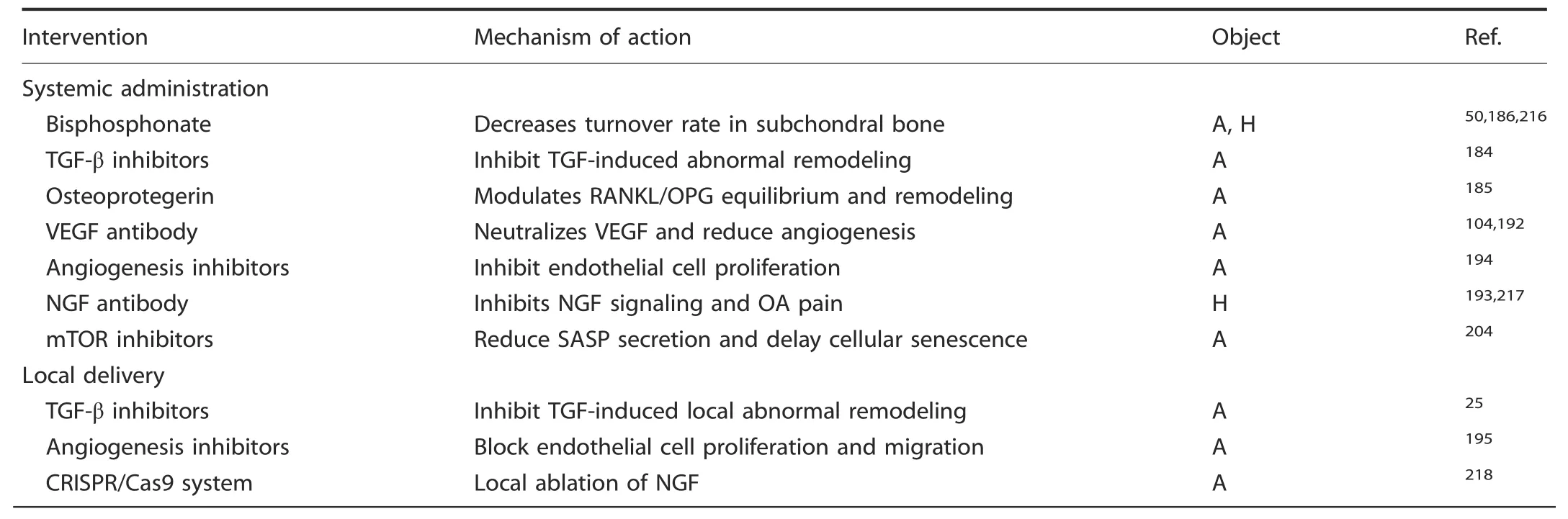
Table 1.Promising pharmacological interventions targeting subchondral bone in OA
Subchondral bone microenvironment regulation
Regulation of uncoupled bone remodeling.High-level bone remodeling has been proven to be responsible for mechanically induced OA,and the inhibition of osteoclast-induced bone resorption could significantly attenuate OA symptoms in a mouse model.TGF-β and osteoclast overactivation have been observed at the onset of OA,and OC-induced matrix resorption increases TGF-β release and activation.The local neutralization of TGF-β in subchondral tissue by knocking out its receptor on MSCs reverses aberrant bone remodeling and excessive angiogenesis,thus attenuating cartilage degradation in ACLT mouse models.25In addition,the effective ingredient of a traditional Chinese medicine,halofuginone,has shown a therapeutic effect in OA patients after systemic delivery by downregulating TGF-β signaling,decreasing bone remodeling and reducing vascularization.184Interestingly,the systemic administration of OPG presented a cartilage-protective effect in a medial meniscectomy model,while OPG did not affect chondrocytes ex vivo,185indicating the OPGinduced inhibition of osteoclast-induced bone remodeling.Runx-2 overexpression-induced excessive bone resorption leads to instability in the hind limbs,and these effects can be rescued by pamidronate,a well-known bone remodeling inhibitor.186The bone turnover rate of subchondral bone has been reported to be both increased and decreased in various studies.Recent ideas interpret these observations as representing different temporal stages of OA progression,which indicates that there is a relatively low remodeling rate and a reduced level of osteopenia in the early stage that initiate a rapid remodeling process and cause subsequent OA pathologies.Otherwise,novel biomaterials or medications precisely targeting osteoclasts187–189and bone remodeling190–191have great potential in local interventions for subchondral bone.In conclusion,the legitimate control of subchondral bone remodeling at appropriate times and locations holds great therapeutic potential.
Inhibition of aberrant angiogenesis.A positive feedback loop has been identified in bone-vascular-cartilage crosstalk during OA involving chondrocytes,mTORC-1,VEGF-A and type H vessels,and the inhibition of angiogenesis by bevacizumab,a VEGF-A antibody,can terminate this feedback loop and thus rescue OA progression.104,192–193Blocking VEGF-induced angiogenesis has shown a significant suppressive effect on vascular invasion and cartilage destruction.Other antiangiogenic factors,such as PPI-2548,194TSP-1,195Chm-1196and angiostatin,197–198have presented similar cartilage protective effects in multiple studies.To make room for new tissue,the matrix of bone or cartilage must be eroded before vessel growth can occur.This process,which is mostly induced by MMP-2,MMP-7 and membrane type 1 matrix metalloproteinase(MT1-MMP),199allows endothelial cells to migrate and further degrade the cartilage matrix,while the inhibition of MMP signaling decreases cellular migration and angiogenesis.200In addition,MMP releases VEGF in multiple matrices and thus accelerates angiogenesis in various diseases,including tumor invasion.201The extracellular domain of FGF receptor type 1 in the vascular basement membrane can be hydrolyzed by MMP-2 to block the intracellular signaling pathway,202thus inhibiting vascularization.In addition to angiogenic factors,several antiangiogenic molecules are also released by MMPs,such as endostatin,arrestin,canstatin and tumstatin,which are derived from various types of collagen.Overall,while antiangiogenic factors have a great latent capacity for OA therapy,further research is needed to clarify the exact timing and methods for the use of MMPs and their inhibitors in treating OA.
Targeting senescence.The selective removal of senescent cells has become a hot spot of research in OA therapeutics.Using drugs to block the antiapoptotic ability of senescent cells and their ability to secrete proinflammatory factors has also been explored as a method to remove senescent cells.In addition to inflammatory factors and excessive wear,senescent cells isolated from the cartilage and synovial tissues of OA patients have been found to contribute to OA progression.142In p16-3MR transgenic mice(an animal model of aging in which p16lnk4a-positive senescent cells can be monitored),researchers injected ganciclovir(GCV)to induce apoptosis and selectively remove senescent cells.In addition,they tested a recently discovered senolytic(UBX0101)that could selectively remove senescent cells,and the results showed a clearing effect on senescent cells.In addition,the senescence of chondrogenic progenitor cells(CPCs)hinders the formation of chondrocytes,and senescent CPCs show an increase in IL-1β synthesis.In an intermittent hydrostatic pressure(IHP)mouse model,the senolytics dasatinib and quercetin were used to selectively induce the apoptosis of aging CPCs in vivo and in vitro,which might promote cartilage formation.203Oxidative stress is one of the most important reasons for the formation of senescent cells,so reducing oxidative stress may be a way to eliminate senescent MSCs.Although the mechanism of cellular senescence in OA is not completely clear,the selective removal of senescent cells,especially in subchondral bone in joints,attenuates the development of posttraumatic OA,which provides new insights into treatments targeting senescent cells in OA.142
At present,there are also cytokine-neutralizing antibody therapies for use against senescent cells.Rapamycin is an mTOR inhibitor,and treatment with rapamycin could reduce the expression of MMP-3,MMP-13,IL-1 and IL-6 in rabbit annulus fibrosus stem cells(AFSCs),inhibit mTOR signaling,reduce SASP secretion,regulate the inflammatory response in vivo,and inhibit cell senescence,thus showing potential for the treatment of agerelated diseases,204including OA.
Chronic pain management
Since OA pain is caused by multiple pathogenic processes,including nociceptive and neuropathic mechanisms,and appears to be heterogeneous,patients of different subtypes should be treated differently.205Large numbers of pain-relieving drugs or bioactive constituents have been reported in recent years,but there is no single medication that works for all subtypes of OA based on present clinical experience.
NSAIDs are recommended as first-line agents for knee OA pain in the AAOS guidelines(2nd edition).206Traditional nonselective COX inhibitors,such as indomethacin and ibuprofen,have substantial side effects in the alimentary canal.Selective COX-2 inhibitors,such as celecoxib and etoricoxib,present a significantly lower risk of gastrointestinal events without decreasing cardiovascular safety.207–208COX-2 acts as a rate-limiting enzyme in osteoblasts,and the inhibition of COX-2 or PGE2-EP4 signaling in subchondral bone results in a tendency toward adipogenesis during MSC differentiation,thus decreasing osteogenesis and angiogenesis and alleviating OA pain.Nonopioid central analgesics,such as tramadol,have been applied in OA pain management for years.The additional administration of tramadol was recommended for senile OA patients who were refractory to NSAIDs in a clinical retrospective study.209In addition,some opioid medications,such as transdermal buprenorphine,have been applied in clinical practice and have achieved good results.210–211
The intraarticular injection of corticosteroids for the treatment of OA pain seems to be common in clinical practice and is aimed at reducing inflammation inside the joint space,but it has been widely questioned in recent years.Research by Bruno et al.212has suggested that intraarticular corticosteroids are associated with a moderate improvement in pain,but this is based on low-quality evidence.When compared with the injection of saline,the local injection of triamcinolone for 2 years resulted in significantly greater cartilage loss and no improvement in pain management.213Patients who received physical therapy exhibited a painless and well-functioning joint,unlike patients who received intraarticular glucocorticoid injections.214In addition,intraarticular hyaluronic acid therapy is not supported or discouraged in the AAOS guidelines206or a report by Grace et al.215
CONCLUSION AND PERSPECTIVE
Complicated anatomical structures and interactive pathologies increase the difficulty of the clinical diagnosis and pharmaceutical treatment of OA.The intricate regulatory mechanisms between cartilage and subchondral bone include both mechanical and biological factors and play critical roles in the onset and progression of OA.Subchondral bone lies subjacent to the cartilage layer and provides mechanical and nutritional support for the cartilage.Multiple studies have reported that asymptomatic subchondral bone marrow lesions present earlier than OA pain or cartilage destruction.Recent research elucidating bone-cartilage crosstalk and subchondral bone metabolism provides a better understanding of this complex of whole-joint disorders.In this review,we summarized the recent progress in research on OA with a focus on subchondral bone.Briefly,abnormal mechanical loading leads to increased subchondral bone turnover,while angiogenesis and nerve innervation facilitate chondrocyte hypertrophy and cartilage degradation.These pathologies in turn accelerate bone remodeling beneath the tidemark,and finally,aberrant mechanical support due to subchondral bone sclerosis induces cartilage destruction.In conclusion,the inefficiency of therapies for local cartilage repair or inflammation inhibition(including arthroscopic debridement and injections),along with the promise of methods for regulating the subchondral microenvironment,suggests a new orientation for OA management in the future.
ACKNOWLEDGEMENTS
We thank Springer Nature Author Services for language editing.This work was supported by the National Key R&D Program of China(2018YFC2001500),the National Natural Science Foundation Key Research Program in Aging(91749204),and the National Natural Science Foundation of China(81771491,81871099).
ADDITIONAL INFORMATION
Competing interests:The authors declare no competing interests.
- Bone Research的其它文章
- Spatially defined single-cell transcriptional profiling characterizes diverse chondrocyte subtypes and nucleus pulposus progenitors in human intervertebral discs
- PGE2/EP4 skeleton interoception activity reduces vertebral endplate porosity and spinal pain with low-dose celecoxib
- Lipocalin 2 stimulates bone fibroblast growth factor 23 production in chronic kidney disease
- TAZ inhibits osteoclastogenesis by attenuating TAK1/NF-κB signaling
- Hepcidin contributes to Swedish mutant APP-induced osteoclastogenesis and trabecular bone loss
- H3K36 methyltransferase NSD1 regulates chondrocyte differentiation for skeletal development and fracture repair

