Therapeutic efficiency of bone marrow-derived mesenchymal stem cells for liver fibrosis: A systematic review of in vivo studies
Zaid Al-Dhamin, Ling-Di Liu, Dong-Dong Li, Si-Yu Zhang, Shi-Ming Dong, Yue-Min Nan
Abstract Although multiple drugs are accessible for recovering liver function in patients, none are considered efficient. Liver transplantation is the mainstay therapy for end-stage liver fibrosis. However, the worldwide shortage of healthy liver donors, organ rejection, complex surgery, and high costs are prompting researchers to develop novel approaches to deal with the overwhelming liver fibrosis cases. Mesenchymal stem cell (MSC) therapy is an emerging alternative method for treating patients with liver fibrosis. However, many aspects of this therapy remain unclear, such as the efficiency compared to conventional treatment, the ideal MSC sources, and the most effective way to use it. Because bone marrow (BM) is the largest source for MSCs, this paper used a systematic review approach to study the therapeutic efficiency of MSCs against liver fibrosis and related factors. We systematically searched multiple published articles to identify studies involving liver fibrosis and BM-MSC-based therapy. Analyzing the selected studies showed that compared with conventional treatment BM-MSC therapy may be more efficient for liver fibrosis in some cases. In contrast, the cotreatment presented a more efficient way. Nevertheless, BM-MSCs are lacking as a therapy for liver fibrosis; thus, this paper also reviews factors that affect BM-MSC efficiency, such as the implementation routes and strategies employed to enhance the potential in alleviating liver fibrosis. Ultimately, our review summarizes the recent advances in the BM-MSC therapy for liver fibrosis. It is grounded in recent developments underlying the efficiency of BM-MSCs as therapy, focusing on the preclinical in vivo experiments, and comparing to other treatments or sources and the strategies used to enhance its potential while mentioning the research gaps.
Key Words: Bone marrow; Mesenchymal stem cells; Liver fibrosis; In vivo; Efficiency
INTRODUCTION
The worldwide incidence of liver fibrosis has been increasing steadily in recent years; even though antiviral agents are widely used, the ultimate treatment for liver fibrosis is liver transplantation. Because transplants are not available in many countries and when they are, high costs and organ shortages cause this to be an unfeasible option for many patients. Development of curative therapies for end-stage liver disease is a necessity. Stem cell transplantation represents a promising solution because it involves recovery of the liver and production of hepatic stem cells (HSCs) in sufficient quantities to overcome the shortage of liver donors. Hepatocyte expansion from HSCs facilitates both physiological turnover and homeostatic regeneration[1].
Mesenchymal stem cells (MSCs) boast the benefits of being acquired relatively easily and stimulating low immunogenicity[2]. These cells are also characterized by a self-renewal ability and a capacity to differentiate into cells of various lineages, including osteoblasts, adipocytes, and chondrocytes[3]. MSCs also elicit less of an ethical concern because they do not originate from somatic cells. Their transplantation is considered safe and has been widely assessed in clinical settings and various diseases, yielding promising results[4].
Because bone marrow (BM) is the largest supplier of MSC sources, we focused this work on BM-MSC transplantation for liver fibrosis and its therapeutic efficiency. Many elements affect the therapeutic efficacy of BM-MSCs, such as culture method, strategies, and transplantation routes. Thus, understanding liver regeneration through BM-MSCs is crucial to offer new perspectives for treatment of liver diseases. This includes the underlying therapeutic mechanism that facilitates alleviation of liver fibrosis, its efficiency compared to other treatments, or dependence on the transplantation route and the strategies used for the procedure. This review outlines the recent advances of BM-MSCs for liver fibrosis, the main aspects of its utility steps, and their therapeutic effects on liver fibrosis to address questions regarding efficacy and gaps in the knowledge, opening a new path toward further studies (Figure 1).
LIVER FIBROSIS
Liver fibrosis is the extreme accumulation of extracellular matrix (ECM) proteins, including collagen, and appears in most chronic liver diseases. Distinct types of hepatotoxic agents produce mediators that induce inflammatory actions in hepatic cell types. Following chronic liver injury, symptoms associated with advanced hepatic fibrosis will appear. When advanced, liver fibrosis results in cirrhosis, liver failure, and portal hypertension, often requiring liver transplantation[5]. Alternatively, it can be resolved if the underlying cause is removed or through the use of an antifibrotic drug or cell therapy (Figure 2). It is possibly a reversible response that resulted from either hepatic insults generated by different chronic diseases, such as nonalcoholic fatty liver disease[6]or repetitive chronic liver injury induced by hepatitis, fat deposition, and continued alcohol consumption[7,8]; for both, the liver may accumulate aberrant myofibroblasts and ECM thus generating liver fibrosis. Depending on the inducing liver disease, liver fibrosis pathogenesis differs; for example, schistosomiasis induces liver fibrosis by accumulating parasitic ova and periocular granulomas in portal veins[9]. Wilson’s disease (or hepatolenticular degeneration), caused by a mutation in the Wilson disease protein (ATP7B) gene, frequently induces liver fibrosis[10]. Furthermore, it has recently been found that metabolic syndromes, including obesity, insulin resistance, and diabetes, are closely related to end-stage liver fibrosis[11].

Figure 1 Introduction of the main parts of the review. First, bone marrow-derived mesenchymal stem cells (BM-MSCs) are introduced as a therapy for liver fibrosis, then the steps before transplantation (culture, strategies, and the choice of transplantation route) are discussed. After that, the efficiency of BM-MSCs for liver fibrosis in vivo are explained through studies that research the efficiency, studies that compare the therapy to other medication sources, and the strategies to enhance the therapeutic efficiency.
Physiologically speaking, liver fibrosis is a healing response to liver injury. It is characterized by excessive deposition of ECM proteins as an outcome of different chronic liver diseases, including viral hepatitis and alcoholic or nonalcoholic steatohepatitis[5,12]. Liver fibrosis is beneficial at first because it can encapsulate the injury and is considered a reversible process at this stage[13,14]; however, it ultimately develops into advanced fibrosis or cirrhosis, which might be irreversible and impairs liver function that leads to subsequent morbidity and mortality[15].
After a severe liver injury, parenchymal cells regenerate and substitute the necrotic or apoptotic cells associated with an inflammatory response and an incomplete ECM deposition. The liver regeneration fails if the hepatic injury persists, and hepatocytes are replaced with abundant ECM containing fibrillar collagen. Depending on the origin of the liver injury, the distribution of this fibrous material differs. In chronic cholestatic disorders and chronic viral hepatitis, the fibrotic tissue is first located around portal tracts. In alcohol-induced liver disease, it is instead situated in pericentral and perisinusoidal areas[16]. Liver fibrosis is related to significant alterations in the quantity and composition of ECM[17]. In advanced stages, the liver holds about six times more ECM than ordinary, including collagens (I, III, and IV), fibronectin, undulin, elastin, laminin, hyaluronan, and proteoglycans[18]. Decreased activity of ECM-removing matrix metalloproteinases (MMPs) is mainly related to the overexpression of their inhibitors (i.e. TIMPs)[12].

Figure 2 The transition from a healthy liver to liver fibrosis. Different types of hepatotoxic agents produce mediators that induce inflammatory actions in hepatic cell types. Following chronic liver injury, symptoms associated with advanced hepatic fibrosis will appear. This can either lead to liver cirrhosis, liver failure, and portal hypertension or can be resolved under the conditions mentioned. HCV: Hepatitis C virus; LPS: Lipopolysaccharide.
Succeeding a chronic injury, HSCs, the primary ECM-producing cells[19], activate and transdifferentiate into myofibroblast-like cells, acquiring contractile, proinflammatory, and fibrogenic properties[20,21]. The activated HSCs accumulate at the spots of tissue repair, discharging significant ECM amounts and regulating ECM degradation. Kupffer cells (KCs) are the primary producer of PDGF, which is the main mitogen for activated HSCs. At the transcriptional and posttranscriptional levels, collagen synthesis in HSCs is regulated[22].
There is a complex interplay among different hepatic cell types the occurs during hepatic fibrogenesis. Many hepatotoxic agents can damage hepatocytes[23]; these damaged hepatocytes release reactive oxygen species (ROS) and fibrogenic mediators and induce white blood cell recruitment via inflammatory cells. Apoptosis of damaged hepatocytes stimulates the fibrogenic actions of liver myofibroblasts[23]. Inflammatory cells activate HSCs to secret collagen, emit inflammatory chemokines, and modulate lymphocyte activation[24,25]. Consequently, a vicious circle of inflammatory and fibrogenic cells stimulating each other occurs[26]. Fibrosis is affected by different T helper subsets, with the Th2 response being associated with more active fibrogenesis[27]. KCs play a main role in liver inflammation by releasing ROS and cytokines[28,29]. Also, changes in the composition of the ECM can directly promote fibrogenesis. Fibrinogen, type IV collagen, and urokinase-type plasminogen activator stimulate resident HSCs by activating latent cytokines, such as transforming growth factor (TGF)-β1[30]. Fibrillar collagens can attach and stimulate HSCs via the discoidin domain receptor and integrins. Furthermore, altered ECM can act as a reservoir for growth factors and MMPs[31].
BM-MSCS
Identification of MSCs
Modern science has witnessed an essential thrust in stem cell research[32], identifying their presence in limited amounts in adult tissues, such as adipose tissue (ADMSCs)[33,34], umbilical cord (UC) tissue[35], amniotic fluid[36,37], breast milk[38,39], synovium[40], BM-MSCs[41], placental cells[42], dental pulp[43], lung, and liver (both adult and fetal)[44]. They are multipotent cells capable of differentiating into distinct cell groups, such as hepatocytes[45].
The therapeutic eminence index represents the amount of research that has advanced into clinical trials in the last 10 years, based on the Macrin et al[46]study (Figure 3). The eminent sources of adult MSCs are ordered following their therapeutic eminence index and are presented as follows: UC is the most eminent, then comes the placenta, AD, endometrium, dental pulp, and dermis successively, and the least eminent sources are amniotic fluid, synovium, and breast milk. Moreover, the cell types into which the isolated MSCs can differentiate vary, ranging from neurons and enterocytes to osteocytes and chondrocytes[47], etc. Among the eminent MSCs, three primary sources are capable of treating liver disease, namely the BM-MSCs, UC-MSCs, and AD-MSCs. Usually, MSCs derived from these sources express no signi?cant differences concerning the morphology and immune phenotype[47].
According to the research by Liu et al[48], the choice of MSCs should be related to the function and repair potentiality of the liver. Therefore, BM was selected as the best source compared to the three most capable sources to treat liver diseases. Accordingly, we decided to concentrate on BM-derived MSCs; as promising as this therapy can be, there are still many aspects of this therapy that need to be investigated.
Identification of BM-MSCs
As presented in Figure 4, the BM consists of two different cell lineages: the hematopoietic tissue cells and the associated stromal cells[49]. BM contains more than one stem cell population, and these include: (1) Hematopoietic stem cells and endothelial progenitor cells (EPCs) obtained through flow cytometric cell sorting (known as FACS) according to cell surface markers; (2) Side population cells present in the subpopulation as side scatters on FACS plot, owing to their ability to efflux Hoechst 33342 dye; (3) MSCs; and (4) Multipotent adult progenitor cells, which are derived through the characterization of adherent cell populations.
Isolation and expansion of BM-MSCs involve aspiration of the iliac crest followed by separation of the mononuclear cell fraction by density-gradient centrifugation and plating for expansion. BM-MSCs can differentiate into ectodermal cell lineages that include neurons, endodermal cell lineages, such as hepatocytes[50], and mesodermal lineages, such as myocytes, chondrocytes, osteocytes, and adipocytes[51]. Considering this differentiation capacity, several possible applications of BM-MSCs have been suggested, tested, and studied[46].
There is a presence of pluripotency markers in BM-MSCs, suggesting that they can differentiate into cell lineages of all three germ layers. These surface marker expression levels and transcription factors play a significant role in distinguishing the stem cell populations[46]. Besides the regenerative and differentiation potentials of MSCs, the immunosuppressive and immunomodulatory properties are critical to their use in cellular therapy[52].
The potential contribution of BM-MSCs to liver fibrosis is presented in Figure 5. Each of the presented elements is a different mechanism that has a specific role that can alleviate liver fibrosis, such as the higher differentiation of AFP, CK18, and CK19, the activation of HSCs, and the higher mobility of KCs. The most dominant axis seems to be transdifferentiation to a collagen-producing myofibroblast cell population. However, other factors can also show a potential contribution to liver fibrosis.
BM-MSCs are known to express the MHC class I antigen but not the MHC class II antigen[53,54]. The coculture of BM-MSCs and HSCs inhibited the proliferation of HSCs and promoted cell apoptosis of HSCs through downregulating the E3 ligase S phase kinase associated protein 2 level, attenuating ubiquitination and increasing the stability of p27[55]. Moreover, BM-MSCs produce various growth factors and cytokines with anti-inflammatory effects in vitro and in vivo to inverse the fibrotic liver state. As transplantation of MSCs upsurges, the serum levels of vascular endothelial growth factor(VEGF), hepatocyte growth factor (HGF), IL-10, and MMP-9 increase in injured livers[56].
BM-MSCs attenuate hepatic fibrosis in vivo by decreased serum levels of collagen I, collagen IV, type III procollagen, hyaluronic acid, laminin, downregulated liver collagen proportionate area, hepatic hydroxyproline, and liver α-smooth muscle actin (SMA). This improvement is accompanied by reduced hepatic levels of TGF-β1, decreased expression of serum TGF-β1, Smad3, and Smad4 but increased Smad7 expression[57,58]. BM-MSCs significantly ameliorate liver fibrosis in mice via stimulation of interferon-γ and inhibition of lymphocyte proliferation; the BM-MSCs also significantly decreased the number of IL-17 producing Th17 cells and the serum level of inflammatory IL-17 while increasing the serum levels of kynurenine, immunosuppressive IL-10, indoleamine 2,3-dioxygenase, and a number of CD4+ IL-10+ T cells to attenuate liver fibrosis[59].

Figure 3 Eminent sources of adult mesenchymal stem cells and the cells into which they can differentiate. Eminent sources of adult mesenchymal stem cells are ordered through their therapeutic eminence index. Umbilical cord is the most eminent, followed by the placenta, adipose tissue, endometrium, dental pulp, and dermis. The least eminent sources are amniotic fluid, synovium, and breast milk. The cell types, such as neurons, enterocytes, osteocytes, and chondrocytes, into which the isolated mesenchymal stem cells can differentiate are variant.
BM-MSCs are also confronting various challenges to reach clinical application requirements, such as the highly invasive donation procedure, the decline in MSC number and differentiation potential with increasing age, demands of a large number of cells for therapy, heterogeneic character of cell quality, low survival ability after transplantation, the weakening of MSC capacities in two-dimensional (2D) culture, and unclear mechanism of MSC function for disease therapy. An essential need for MSC therapy is to produce enough high-quality MSCs in vitro to meet clinical demand.
PRE-TRANSPLANTATION STEPS
Culture
As explained in Figure 6, different methods are used to culture stem cells; the general way is to culture MSCs in 2D dishes as a monolayer for fast expansion. This method conjures changes in MSCs, including cellular senescence, immunogenicity, losses of their stemness properties and paracrine activity, genetic expression of cells, and altered inner structure of cells[60,61]. The second way is the three-dimensional (3D) culture, which artificially creates an environment in which cells can interact or grow with their surroundings in all three dimensions. Thus, 3D culture is regarded as a more suitable and closer physiological microenvironment for cell growth[62,63]. There are numerous 3D culture methods developed to form MSC spheroids, such as hanging-drop, magnetic levitation, chitosan membrane culture, microgravity bioreactor, and rotating culture[1,64]. These methods provide cells with a suspension culture condition where the 3D spheroids were formed mainly relying on cell-cell adhesion and interaction that promoted the self-assembly tendency of MSCs.

Figure 4 Bone marrow extracted cells. Bone marrow contains a variety of stem cell populations that can be extracted, either through specific growth factor media, such as that for multipotent adult progenitor cells (MAPCs) and mesenchymal stem cells (MSCs) or through flow cytometric cell sorting (FACS) technology, such as for the endothelial progenitor cells (EPCs), hematopoietic stem cells (HPSCs), and side population cells (SPs).
There are two main types of spheroid used for the 3D culture. The first of these is initially formed and derived from the aggregation of many individual cells and is named multiple cells-derived spheroid (MCDS). Growing evidence has shown that, in comparison to 2D culture, 3D MCDS culture enhances the characteristics of MSCs on cell survival, factor secretion, stemness maintenance, migration, and antisenescence in vitro and improves the capacities of anti-inflammation, angiogenesis, tissue repair, and regeneration in vivo[65,66]. However, despite the many advantages reported, visible defects restrict the direct application of MCDS-cultured MSCs in the clinic. These include the heterogeneity of cell quality in the whole spheroid, the multitudinous presence of individual MSCs with distinct viabilities, and the large size of MCDS resulting in different distributions of nutrients, oxygen, and waste metabolism between the core and periphery of the spheroid; moreover, the cells in the core are subjected to hostile metabolic stresses and tend to undergo apoptosis[67]. The large size (diameter > 100 μm) makes MCDSs unable to be directly injected into the body, as it poses risk of blood vessel blockage. So, the MCDSs generally must be dissociated into single cells by an enzymatic process before vein injection, but this affects the cells by causing damage and impairing viability[68].
The second type of spheroid is formed through a single cell-derived sphere (SCDS), based on the report by Qiao et al[69]. This formation can enhance the effectiveness of UC-MSCs thereby optimizing the quality of MSCs to meet the demand of the clinical application. In vitro and in vivo results have indicated that compared to 2D and MCDS cultures SCDS culture possesses some advantages for MSCs optimization, such as in cell stemness properties, survival ability, and therapeutic potential. However, despite this, there are still some questions that need to be explored further in the future; in particular, these questions involve the effects of SCDS culture on immunomodulatory capacities, inflammatory response, paracrine capacities, and cellular metabolism. Whether SCDS culture could markedly optimize BM-MSCs for potentially meeting the demand for clinical application also remains an unanswered question. In general, after cell transplantation, only a small number of MSCs migrate to injured tissues, so various studies have investigated effective strategies for improving the survival rate and activity of MSCs to treat liver fibrosis.

Figure 5 Potential contributions of bone marrow-derived mesenchymal stem cells to liver fibrosis. Each of the presented elements represent a distinct mechanism that has a specific role that can contribute to alleviating liver fibrosis. BM-MSCs: Bone marrow-derived mesenchymal stem cells; HSCs: Hepatic stellate cells; KC: Kupffer cells.
Strategies to improve MSCs efficiency
BM-MSCs have limited viability, with as low as < 1% of transplanted cells predicated to survive. Inflexibility of the microenvironment encountered upon transplantation may be the cause[70]. Various strategies have been developed and implemented to improve cell therapy. In this section, we will focus on: genetic engineering and the preconditioning used during the culture phase; tissue engineering used on a 3D matrix and involving signaling molecules; and cell-free therapy achieved through the use of exomes (Ex) and microvesicles (MVs) (Figure 7).
Genetic engineering:BM-MSCs have also been genetically engineered to overexpress the desired gene to improve their therapeutic efficacy further. They can be used for the targeted delivery of therapeutic gene products as gene therapy. The genes capable of manipulation could be genes encoding receptors, growth factors, and cytokines. Genetically-engineered BM-MSCs have been applied as treatment to a range of genetic and acquired diseases. Genetic modification of BM-MSCs improves their therapeutic potential by enhancing various cellular features, like endurance and survival of the transplanted BM-MSC, angiogenesis, differentiation, homing, and anti-inflammatory effects[71].

Figure 6 Two main types of mesenchymal stem cell culture. The two-dimensional (2D) culture using 2D dishes as a monolayer for fast expansion, and the three-dimensional (3D) culture with two main types of spheroid use: multiple cell-derived spheroid and single cell-derived sphere. MSCs: Mesenchymal stem cells.
This strategy investigated approaches to promote the expression of proteins involved in the homing of donor cells[72]. MSCs express low levels of molecules, including the homing factor stromal cell-derived factor-1 (SDF-1) and chemokine receptors[73]. Genetic manipulation of prosurvival or antiapoptosis genes have been shown to increase BM-MSC survival in vivo[74]. Through modulation of cellular networks, microRNAs can regulate mRNAs, including those involved in cell survival. MicroRNA overexpression can enhance BM-MSC survival[75]. Nonetheless, this strategy presents many risks, including carcinogenesis, that should be carefully considered when applying genetic manipulations.
Tissue engineering:Strategies that allow for BM-MSC homing and adaptation in the liver before initiating their regeneration will help improve cell survival. Several approaches have been investigated, involving coculture and the development of 3D systems that can involve a scaffold-based or scaffold-free system[76,77]. Cells grown in 3D systems would behave more like cells in vivo and could be implanted directly. Numerous synthetic polymers as well as natural materials have been assessed for their ability to raise the expression of hepatocyte-specific genes in BM-MSCs through hepatic differentiation[78]. The most significant performance effect was observed when a 1:5 ratio of BM-MSCs to hepatocytes was used both in vitro and in vivo[79]. Decellularized tissue is another system in use for tissue engineering; the decellularized liver tissue forms an ECM scaffold, improving MSC engraftment by offering a more physiological environment[80].
Preconditioning:Priming methods avoid genetic and chemical modifications entirely by altering culture conditions to influence gene expression[81]. These methods have been used to improve the tethering, activation, and transmigration steps of systemic homing. Preconditioning improves the survival signals and resistance of MSCs against stress and insults in the pathological environment[82]. In the preconditioning process, BM-MSCs can be pretreated or exposed to a sublethal dose of various insults, such as apoptotic cascade activation, hypoxia, toxins, ROS, inflammatory response, autophagy, and many others. Furthermore, preconditioning can enhance cell survival following the transplantation because it considerably induces therapeutic benefits of BM-MSCs by increasing the potential of cell differentiation and its paracrine protective effect, improving migration and homing of BM-MSCs to the lesion site, increasing regenerative and repair potentials, and suppressing inflammatory and immune responses that occur after transplantation[83]. Many preconditioning strategies involve exposing cells to a physical or an environmental shock and/or pharmacological modulators of targeted molecules[83,84]; the following three strategies exemplify such.
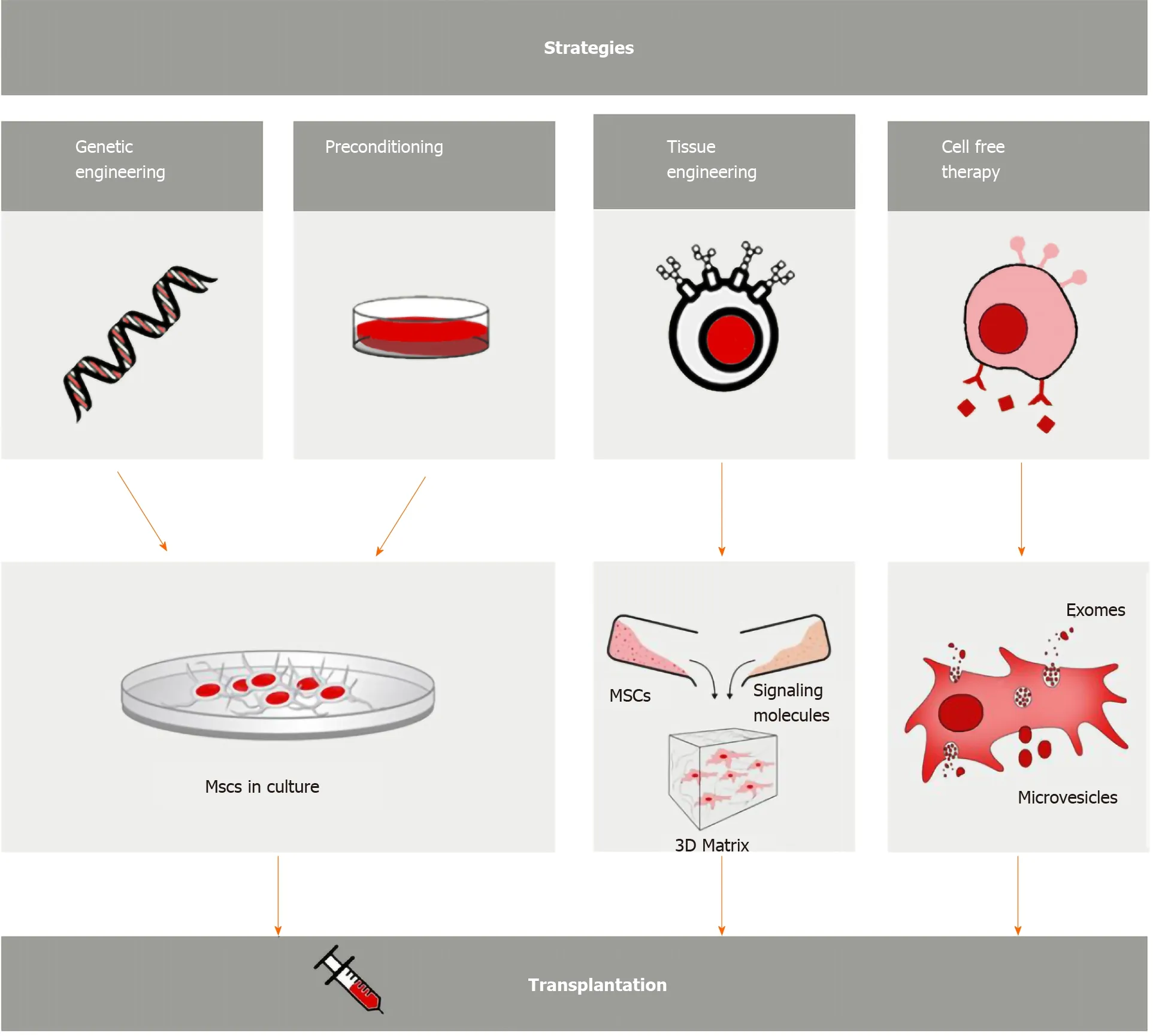
Figure 7 Strategies to enhance bone marrow-derived mesenchymal stem cell therapeutic efficiency. Genetic engineering and preconditioning used during the culture phase; tissue engineering used on a three-dimensional (3D) matrix and involving signaling molecules; cell-free therapy through the use of exomes and microvesicles. MSCs: Mesenchymal stem cells.
The first is a thermal preconditioning strategy carried out at 42 °C for 1-2 h before transplantation. It has been demonstrated to promote cell survival in vivo, and this outcome is related to the induction of heat shock protein expression, which inhibits apoptotic pathways[85,86].
The second is a hypoxic preconditioning strategy based upon the knowledge that hypoxia can promote defense mechanisms against oxidative stress. Hypoxia is a significant feature of MSCs; it plays a vital role in maintaining stem cell fate, selfrenewal, and multipotency. Cultivating MSCs under hypoxia is an essential preconditioning step because it mimics the natural microenvironment of BM. The reaction of MSCs to hypoxic conditions is contradictory, however, indicating both damaging and ameliorating effects.
The third is a pharmacologic strategy to maintain cell viability after transplantation. This process includes the use of antioxidants and HIF-1α stabilizers to contribute to cell survival, as well as antimycin and mitochondrial electron transport inhibitors to promote cell survival by activating mitochondrial death pathways[87].
Extracellular vesicles as a cell-free therapy
Worries regarding the use of MSCs as a cellular therapeutic approach for the liver include their potential for aberrant differentiation, the peril of tumor formation, and the half-life of transplanted MSCs inadequate for tissue regeneration by differentiation[88]. To deal with these issues, the MSC secretome has been introduced as an acellular alternative therapy. Indeed, these soluble proteins or extracellular vesicles residing among the BM-MSCs and released by paracrine mechanisms could be a practical option and offer numerous advantages compared to the use of cellular therapies for liver diseases[89].
BM-MSCs can also release more elaborate structures, called extracellular vehicles (EVs)[90]. These EVs can be engineered to enhance anticipated activities or introduce specific effector molecules[91,92]. MSC-derived EVs were shown to improve hepatic injury and inflammation[93]. EVs from human MSCs preserve at least some of the immunomodulatory properties of the cells. MSC-derived induced pluripotent stem cell-EVs hold the EV characteristics that are usually obtained from tissue-derived MSCs, regardless of origin[94]. EVs could be a better therapeutic strategy because they characterize a physically different fraction and transport signals with more predictable effects. Although, the complex functions of EVs are still mostly undiscovered. Additional studies are needed to determine how long-circulating MSC-EVs survive after administration and what recognition pathways are used by the target cells.
Choice of transplantation routes
Many BM-MSC transplantation routes can be used for liver disease, in general. Some of the routes are direct, such as the portal vein and the hepatic artery; others are indirect routes, such as the peripheral vein, intrasplenic, intraperitoneal, BM reconstitution, and extra-corporeal liver assist device (Figure 8).
BM-MSC transplantation routes can affect the therapy’s potential because they conceptually represent the simplest method to improve MSCs homing by administrating the cells at or near the target tissue instead of infusing them through standard intravenous routes. It may seem intuitive that direct delivery of MSCs to a target tissue could result in higher retention[95].
There are conflicting data about the engraftment of transplanted BM-MSCs, and some concerns around the fibrogenic potential have been raised. Unwanted effects may depend on the route and dose of BM-MSC infusion[96,97]. Different routes can be used to transplant BM-MSCs, such as intravenous, intraperitoneal, intrahepatic, intrasplenic, or portal vein injection, but the effectiveness varies depending on the injection route. The peripheral vein is the most common transplantation route followed by the hepatic artery, intrasplenic, intrahepatic and portal vein injection. It has been shown that BM-MSCs administered through the peripheral vein migrate well into the liver parenchyma in chronic injury in vivo. Simultaneously, limited BM-MSC engraftment has been observed in an acute injury environment[98].
Furthermore, BM-MSCs endured in liver tissues when injected through the intrahepatic artery, indicating that BM-MSCs were present without differentiating into hepatocytes. Moreover, the intraportal infusion was found to be more efficient than the peripheral route in clinical trials. However, direct approaches, such as via the portal vein or hepatic artery, carry a risk of portal hypertensive bleeding following cell injection[99]. Generally, the lines of evidence provided by most of these clinical studies have been lacking.
EFFICIENCY OF BM-MSCS FOR LIVER FIBROSIS IN VIVO
Several in vivo studies have continued to prove the efficiency of BM-MSCs in attenuating liver fibrosis induced by tioacetamide or carbon tetrachloride. These studies have investigated the efficiency and role of MSCs in liver fibrosis to elucidate the mechanism underlying the mobilization and function of BM-MSCs.
Mehrabani et al[100]investigated the regenerative effect of BM-MSCs in a rat model of liver fibrosis induced by tioacetamide. The study demonstrated that BM-MSCs could open a new window and be a therapy of choice in the amelioration of liver fibrosis because it alleviated liver fibrosis through the antifibrotic potential of BM-MSCs. The paracrine and endocrine functions of BM-MSCs also underlay the efficacy of these cells in the amelioration of liver damage through reducing inflammatory cells in the hepatic tissue and decreasing the alanine aminotransferase level.
Another study investigated the link between natural killer cells and liver fibrosis and their link to regenerative medicine. It was found that BM-MSCs alleviated liver fibrosis through suppressing the inflammatory response and the local proinflammatory cytokines. A significant increase in intrahepatic natural killer cells was also noted upon BM-MSC treatment[101]. BM-MSCs attenuate liver ?brosis by decreasing serum levels of the in?ammatory cytokine IL-17, increasing the immunosuppressive cytokine IL-10 and the related factors indoleamine 2,3-dioxygenase and kynurenine, reducing the number of IL-17-producing Th17 cells, and expanding the percentage of CD4+ IL-10+ T cells[59].

Figure 8 Bone marrow-derived mesenchymal stem cell transplantation routes for liver disease. The routes are either direct (portal vein and hepatic artery) or indirect (peripheral vein, intrasplenic, intraperitoneal, bone marrow reconstitution, and extra-corporeal liver assist). BM-MSCs: Bone marrow-derived mesenchymal stem cells.
The BM-MSCs have also been shown to improve thioacetamide-induced liver fibrosis in rats by remolding the collagen ?bers, which could be lysed by MMPs, namely MMP-2-mediated degradation of the ECM. PKH26-labeled BM-MSCs were seeded into liver tissue and found to differentiate into healthy cells replacing the damaged ones with either hepatocytes or cholangiocytes. The reduction in α-SMA expression that was observed re?ected a diminution in the number of activated HSCs[102].
BM-MSCs might play an immunomodulatory role in treating liver fibrosis through the down-regulation of IL-17A, affecting the IL-6/STAT3 signaling pathway. It was noted that after the treatment there was modulation of the cytokine milieu and among the signal transducers, including a significant downregulation of the genes encoding cytokines IL-17A, IL-17RA, IL-17F, and IL-17RC. In accordance with the BM-MSC administration was a decline in IL-17, IL-2, and IL-6 serum proteins and downregulation in the IL-17A and IL-17RA proteins in liver tissue. The BM-MSC administration also resulted in downregulation of both Stat3 mRNA expression and p-STAT3 protein as well as Stat5a gene expression and p-SMAD3 and TGF-βR2 proteins and elevated p-STAT5 protein[103].
It was also evidenced that the SDF-1α/CXCR4, which is essential among the chemotactic axis regulating MSC migration from BM to fibrotic liver, can attenuate liver damage and thus alleviate liver fibrosis[104]. That same study identified VEGF as the key cytokine that contributes to MSC proliferation. These results provide further evidence for the role of MSCs in liver fibrosis and help to elucidate the mechanism underlying MSC mobilization under the condition of carbon tetrachloride-induced liver injury (the model system used in the study).
Transplantation of stem cells, including BM-MSCs, has proven to be competent for repairing fibrotic livers. The underlying mechanism promotes hepatocyte transdifferentiation and hepatocyte proliferation while inhibiting activated hepatic stellate cells, upregulating the activity of MMPs, and promoting neovascularization in liver tissues[105].
Considering that BM-MSCs have demonstrated a strong proliferative ability, multilineage potential, and no ethical considerations for widespread application to repair various organ injuries, they are currently transplanted in vivo to reduce hepatocyte apoptosis and promote hepatocyte regeneration[106]. Thus, in this review, we focused on the in vivo research in the literature starting from the year 2015 to address these two main issues: (1) The therapeutic efficiency of BM-MSCs compared to other types of treatments; and (2) The possibility to enhance the therapeutic efficiency of BM-MSCs through various strategies.
Therapeutic efficiency of BM-MSCs compared to other treatments and stem cell sources
The therapeutic efficiency of BM-MSCs depends on various factors, but there remains a need to compare this efficiency to that of other treatments and other MSC sources. In Table 1[107-116], we present those studies in the literature that have compared BM-MSC efficiency to other existing therapies and other sources up to the year 2015.
We noted that compared to such standard treatments as resveratrol and silybum marianum the regenerative capabilities and resolution of hepatic fibrosis was higher for the BM-MSCs. Treatment with BM-MSCs enhanced the liver state more effectively than either of the two drugs. It also significantly decreased levels of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, malondialdehyde, TNF-α, and CYP450 and increased levels of albumin, superoxide dismutase, glutathione, glutathione S-transferase, and catalase. BM-MSCs could also reestablish liver structure and function, ameliorating the toxicity of carbon tetrachloride and improving liver function tests[107].
In other cases, drug treatment with imatinib, simvastatin, and decorin was found to be more efficient when used concomitant to BM-MSCs. For example, with imatinib the single treatment and combination therapy significantly reduced serum levels of alanine aminotransferase, aspartate aminotransferase, and alkaline phosphatase and downregulated α-SMA, procollagen I, procollagen III, collagen IV, and laminin. However, in pathological observations the highest therapeutic potential was achieved with a combination of BM-MSCs and imatinib[108]. Also, compared to simvastatin and decorin the cotreatment exhibited better histological improvement than was achieved with simvastatin or decorin alone. The combination treatment also lowered the hydroxyproline content, decreased hepatic collagen distribution, and rescued liver function impairment. The upregulation of α-SMA, collagen-1, TGF-b1, and p-Smad3 was prevented by the administration of the cotreatment, which exerted strong protective effects against hepatic fibrosis[109,110].
Depending on the source, the efficiency of MSCs differ; for instance, compared to human UC CD34+ cells, both the CD34+ cells and BM-MSCs have the same efficacy in significantly reducing TNF-α. Nevertheless, concerning liver function and gene expression, the UC CD34+ cells were more efficient in elevating albumin and reducing alanine aminotransferase concentrations and gene expression of collagen Iα, TGF-β1, α-SMA, albumin, and MMP-9. Thus, human UC CD34+ stem cells were deemed more efficient than the BM-MSCs[111].
While the UC-MSCs present similar effectiveness to BM-MSCs, levels were also compared to UC-EPCs and AD-MSCs. The UC-EPCs showed higher MMP-2 and VEGF gene expression than BM-MSCs. Moreover, the UC-EPCs were more effective than BM-MSCs in increasing gene expression of HGF, α-SMA, and Ki-67. The UC-EPCs also showed significantly higher TGF-β than BM-MSCs[112]. Comparison of BM-MSCs and AD-MSCs showed them to be similarly efficient at attenuating liver fibrosis, both using a mechanism that involves inhibiting the activation and proliferation of HSCs and boosting apoptosis of HSCs. The AD-MSCs may be a better candidate than BMMSCs for cell-based therapy to treat liver fibrosis because they improved the antiinflammatory and antifibrotic effects to a slightly greater extent than the BM-MSCs, they are easier to prepare, and they are more effective at inhibiting HSC proliferationand apoptosis in the coculture system. However, the slight improvement in antiinflammatory and antifibrotic effects did not reach the threshold of statistically significant difference[113,114].

Table 1 Bone marrow-derived mesenchymal stem cell therapeutic efficiency compared to other treatments and other stem cell sources
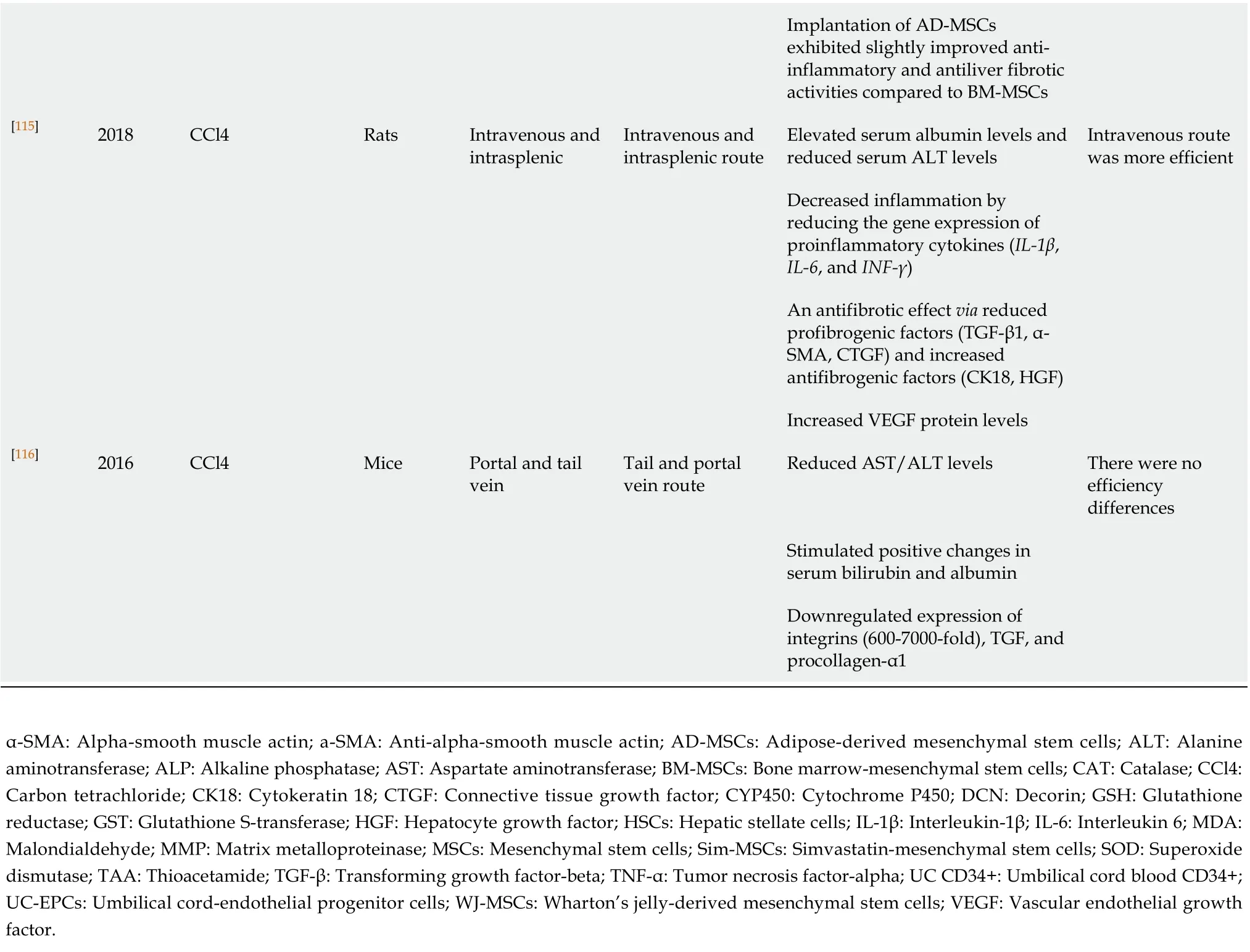
Implantation of AD-MSCs exhibited slightly improved antiinflammatory and antiliver fibrotic activities compared to BM-MSCs[115]2018 CCl4 Rats Intravenous and intrasplenic Intravenous and intrasplenic route Elevated serum albumin levels and reduced serum ALT levels Intravenous route was more efficient Decreased inflammation by reducing the gene expression of proinflammatory cytokines (IL-1β, IL-6, and INF-γ)An antifibrotic effect via reduced profibrogenic factors (TGF-β1, α-SMA, CTGF) and increased antifibrogenic factors (CK18, HGF)Increased VEGF protein levels[116]2016 CCl4 Mice Portal and tail vein Tail and portal vein route Reduced AST/ALT levels There were no efficiency differences Stimulated positive changes in serum bilirubin and albumin Downregulated expression of integrins (600-7000-fold), TGF, and procollagen-α1 α-SMA: Alpha-smooth muscle actin; a-SMA: Anti-alpha-smooth muscle actin; AD-MSCs: Adipose-derived mesenchymal stem cells; ALT: Alanine aminotransferase; ALP: Alkaline phosphatase; AST: Aspartate aminotransferase; BM-MSCs: Bone marrow-mesenchymal stem cells; CAT: Catalase; CCl4: Carbon tetrachloride; CK18: Cytokeratin 18; CTGF: Connective tissue growth factor; CYP450: Cytochrome P450; DCN: Decorin; GSH: Glutathione reductase; GST: Glutathione S-transferase; HGF: Hepatocyte growth factor; HSCs: Hepatic stellate cells; IL-1β: Interleukin-1β; IL-6: Interleukin 6; MDA: Malondialdehyde; MMP: Matrix metalloproteinase; MSCs: Mesenchymal stem cells; Sim-MSCs: Simvastatin-mesenchymal stem cells; SOD: Superoxide dismutase; TAA: Thioacetamide; TGF-β: Transforming growth factor-beta; TNF-α: Tumor necrosis factor-alpha; UC CD34+: Umbilical cord blood CD34+; UC-EPCs: Umbilical cord-endothelial progenitor cells; WJ-MSCs: Wharton’s jelly-derived mesenchymal stem cells; VEGF: Vascular endothelial growth factor.
The transplantation route also impacts BM-MSC competence. This is not the case for all though, as there are no notable differences in the impact from portal and tail vein injections. The former involves different procollagen gene expression than the latter; nevertheless, the liver serum markers and liver histology classification show no differences postinjection. Remarkably, there are also no differences in treatment effects for those two administrations. When considering safety, though, BM-MSC transfusion via a peripheral vein is a safer potential method. In other cases, the impact results differ; for example, the intravenous route is remarkably more efficient than the intrasplenic one. Although both routes achieve a similar enhancement of liver function, the intravenous route provides greater reduction in cytokine gene expression levels (IL-1β, IL-6, and INF-γ)[115,116].
As a result, we note that although BM-MSCs are more efficient than other treatments, combination treatment with other therapies can itself become a strategy to enhance the therapeutic potential of BM-MSCs. We also want to point out that even though BM-MSCs are considered the most significant source of stem cells and are more efficient in some cases, there should be more studies to consider the potential of other sources potential. Lastly, many possible transplantation routes can affect the therapeutic impact of BM-MSCs on liver fibrosis, but we have observed a lack of studies that research the advantages and disadvantages of these routes.
Possibility to enhance the therapeutic efficiency of BM-MSCs
The therapeutic efficiency of BM-MSCs as a treatment for liver fibrosis is influenced by numerous factors including culture conditions, delivery route, number of infused cells, gene modification of MSCs, and other potential factors. Hence, we herein arrange and analyze the current evidence related to BM-MSC transplantation in liver fibrosis and summarize the strategies for promoting the therapeutic efficiency of BM-MSC transplantation. We expect to develop other strategies to improve BM-MSC activities in vivo to restore liver function and alleviate liver fibrosis. Based on Table 2[117-129], theoverall strategies used have a positive therapeutic impact on liver fibrosis. Notable among the various strategies are preconditioning using autophagy regulation, microencapsulation, and preconditioned media.

Table 2 Strategies to enhance bone marrow-derived mesenchymal stem cell therapeutic efficiency

[123]2015 CCl4 Rats Intravenous Genetically modified BM-MSCs expressing TIMP-1-shRNA Decreased TIMP-1 expression thereby regulating HSC survival Decreased serum levels of ALT and AST, fibrotic areas, and collagens Reduction of the fibrotic area Restoration of the liver function[124]2020 CCl4 Mice Intraperitoneal injection MSCs expressing EPO Promoted cell viability and migration of BM-MSCs Enhanced anti?brotic ef?cacy with higher cell viability and stronger migration ability Alleviated liver ?brosis[125]2015 BDL or CCl4 Mice Underneath the kidney capsule Microencapsulated BM-MSCs Activated HSCs Released antiapoptotic (IL-6, IGFBP-2) and anti-inflammatory (IL-1Ra) cytokines Decreased mRNA levels of collagen type I Increased levels of MMPs[126]2018 CCl4 Rats Tail vein Genetically modified BM-MSCs with human MMP-1 Biochemical parameters and hepatic architecture improved Decreased collagen content Suppressed activation of HSCs Improvement of both liver injury and fibrosis[127]2016 CCl4 Rats Tail vein Human urokinase-type plasminogen activator gene-modified BM-MSCs Decreased serum levels of ALT, AST, total bilirubin, hyaluronic acid, laminin, and procollagen type III Genetically modified BM-MSCs with human urokinase-type plasminogen activator Increased levels of serum albumin Downregulated both protein and mRNA expression of β-catenin, Wnt4, and Wnt5a Decreased the Wnt signaling pathway Decreased mRNA and protein expression of molecules involved in Wnt signaling thus working as an antifibrotic[128]2015 TAA Mice Tail vein Genetically modified BM-MSCs, MSCs engineered to produce IGF-I Enhanced the effects of MSC transplantation Decreased inflammatory responses

ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; BDL: Bile duct ligation; BM-MSCs: Bone marrow-mesenchymal stem cells; CCl4: Carbon tetrachloride; EPO: Erythropoietin; hBM-MSCs-Ex: Human BM-MSCs-exomes; HBM: Human bone marrow; HGF: Hepatocyte growth factor; HSCs: Hepatic stellate cells; IGF-I: Insulin growth factor like-I; IL: Interleukin; MMPs: Matrix metalloproteinases; MSCs: Mesenchymal stem cells; MT: Melatonin; MVs: Microvesicles; PGE2: Prostaglandin E2; PPARγ: Peroxisome proliferator-activated receptor-gamma; PTGS2: Prostaglandin-endoperoxide synthase-2; S1P: Sphingosine 1-phosphate; S1PR3: Sphingosine-1-phosphate receptor 3; TGF: Transforming growth factor; TIMP-1: Tissue inhibitor of metalloproteases 1; UTR: Untranslated region; VEGF: Vascular endothelial growth factor; WISP1: Wnt-1-induced secreted protein 1.
BM-MSCs conditioned medium preparation provided a predominant therapeutic role in experimentally-induced chronic liver fibrosis as demonstrated by improved morphological, immunohistochemical, and biochemical measures[118]. Still, future studies should be carried out to further delineate the mechanisms underlying their action.
Cell-free therapy with BM-MSC-MVs and human BM-MSCs-Ex is another method by which the healing effect is enhanced as demonstrated by human BM-MSCs-Ex providing a meaningfully greater therapeutic effect than the human BM-MSCs. In fact, the human BM-MSCs-Ex effectively alleviated liver fibrosis as evidenced by reduced collagen accumulation, inhibition of inflammation, enhanced liver functionality, and increased hepatocyte regeneration. Besides, the administration of hBM-MSCs-Ex reduced liver fibrosis via inhibition of Wnt/β-catenin signaling to prevent HSC activation. Therefore, the use of hBM-MSCs-Ex presents a new and promising therapeutic strategy for hepatic disease in the clinical setting[122].
Adding to that, genetically-modified BM-MSCs expressing TIMP-1-short hairpin RNA or with human MMP-1 boosted the original effects of BM-MSCs through a mechanism that involved enhancing the antifibrotic potential as well as the antiinflammatory and proangiogenic effects. Although BM-MSC administration reduced liver fibrosis, transplantation of the BM-MSCs/MMP-1 enhanced the reduction in liver fibrosis to a greater extent. Therapy with BMSCs/MMP-1 also reduced collagen content and suppressed activation of HSCs in the fibrotic liver, leading to the subsequent improvement of liver injury and fibrosis[126].
In other cases, such strategies as preconditioning with melatonin enhanced the homing and transplantation of the BM-MSCs. Remarkably, melatonin-BM-MSCs showed better therapeutic outcomes, likely facilitated through a mechanism involving the improvement of cell homing and better maintenance of the balance between matrix degradation and accumulating factors that had a high ability to home into the injured liver. This could be related to a higher percentage of glycogen storage but a lower percentage of collagen and lipid accumulation. On the other hand, this could be due to lower expressions of TGF-β1 and Bax, and lower content of serum alanine aminotransferase but higher expressions of MMPs and Bcl2[120].
Simultaneously, other strategies, such as MSCs expressing EPO promoted cell viability and strengthened their migration ability to damaged cells. As an example, compared to BM-MSCs EPO-MSC treatment was found to promote cell viability and migration of the BM-MSCs and to enhance the anti?brotic ef?cacy without inducing apoptosis[124]. This finding supported improving the ef?ciency of MSC transplantation as a potential therapeutic strategy for liver ?brosis.
Overall, each strategy tackles specific aspects that can enhance the therapeutic potential of BM-MSCs, but there are no strategies that tackle several aspects at the same time. Therefore, there is a need to create strategies that can affect various aspects and mechanisms related to improving the potential of BM-MSCs as a therapy for liver fibrosis. Moreover, these strategies need to have sufficient safety, proficiency, and productivity features. It is also important to mention a deficiency in all types of strategies to enhance the therapy’s efficiency. Because there are many undiscovered and unclear aspects of the therapeutic mechanism of BM-MSCs for liver fibrosis, there is also room for new strategies to be discovered and researched.
Limitations and further study
This review successfully studied the recent advances in therapeutic efficiency of BMMSCs for liver fibrosis, focusing on the preclinical in vivo experiments while mentioning the gaps that still exist within the field. However, this review remains limited because it focused mostly on preclinical studies performed on animals; although these studies targeted many aspects of the topic, different targets still need to be addressed. This review did not address clinical studies, but it is crucial to address all the concerns regarding long-term follow-up exams for humans. Only then may we draw solid conclusions on the therapeutic effects of BM-MSC transplantation on liver fibrosis. Ultimately, such additional work will also help to further improve the therapeutic effects of BM-MSC transplantation for liver fibrosis, enhancing the quality of life and prolonging patient survival time with liver fibrosis.
CONCLUSION
Various therapeutic methods are used to alleviate liver fibrosis; BM-MSCs are a promising therapy being investigated in vivo. This type of therapy has diverse advantages, including anti-inflammatory, self-renewal, and multipotency abilities. Thus, more and more studies are investigating the anti-inflammatory and immunomodulatory effects of BM-MSCs and focusing on comparing them with other stem cell sources and treatments to identify and develop an optimal treatment for the regression of liver fibrosis. Ongoing studies are focused on determining the different factors affecting the therapeutic efficiency of BM-MSCs, such as the transplantation route, where the portal vein route may be the optimum choice for restoring liver function in liver fibrosis, and the strategies used, including BM-MSC-based cell-free therapy and preconditioned, tissue-engineered, and genetic-engineered BM-MSC transplantation. These strategies have thus far presented promising results, but more research and experiments need to be done to find the optimum and most efficient strategy (or strategies) to enhance the therapeutic effect and be elevated to clinical trials.
 World Journal of Gastroenterology2020年47期
World Journal of Gastroenterology2020年47期
- World Journal of Gastroenterology的其它文章
- Artificial intelligence-aided colonoscopy: Recent developments and future perspectives
- Invasive fungal infection before and after liver transplantation
- Molecular overview of progressive familial intrahepatic cholestasis
- Evaluation of an educational telephone intervention strategy to improve non-screening colonoscopy attendance: A randomized controlled trial
- Towards an evaluation of alcoholic liver cirrhosis and nonalcoholic fatty liver disease patients with hematological scales
- Prevalence and associated factors of obesity in inflammatory bowel disease: A case-control study
